| II. The Greater Powers of Neolithic Man: the Benefits of Tools, Communication, and Social Organization | Title page | IV. Power and the Machine as Phases of Man’S Harnessing of Nature |
[ p. 71 ]
PART II
MAN, THE HARNESSER OF NATURE: MULTIPLICATION OF MAN’S POWERS
PURPOSES OF PART II
- To show some of the more striking ways in which man fias increased his powers by harnessing nature.
- To shpw why scientific knowledge is such a great aid to man.
- To show how important it is to be able to harness nature if we wish to live together well.
CHAPTER HEADINGS OF PART II
Chapter III. Fire and the Metals as Phases of Man’s Harnessing of Nature.
Chapter IV. Power and the Machine as Phases of Man’s Harnessing of Nature.
Chapter V. Science, the Creative Stage of Man’s Harnessing of Nature.
Chapter VI. Harnessing Nature and Living Together Well.
[ p. 72 ]
Neolithic man lived on the same earth as 'ou and 1, but he lived far more meageiiy. One reason for his meager living was his lack of ability to make the best use of nature’s powers.
Quite unaware of the sleeping giants beneath his feet, he hunted game over the same dejrosits of coal, petroleum, and iron ore that now give us our powerdriven machines. With digging stick or tortoise-shell hoe he scratched the soil that is to-day turned by giant plows drawn by tractors. He put up his miserable shelters on the same spots where to-day we find a country mansion or the skyscrapers of New York or Chicago. He spent days on the same trails that are to-day covered in hours by the locomotive. The same nature that is to-day harnessed by science was, for neolithic man, filled with unknown “spirits” and “magic.”
Evidently man has greatly multiplied his power over nature since the day of neolithic man.
In Chapters III, IV, V, and VI we are to .see some of the main steps by which man has harne.ssed nature and made her do his bidding. In Chapter HI, we shall see how he learned to command fire and the metals; in Chapter IV, how he has harnc,s.sed st,eam, gas, and electricity and put them to turning machines for him; in Chapter V, how he has developed scientific knowledge, that greatest of all harnessers, and has forced natme to tell some of her amazing secrets. These secrets turned out not to be magic at all! In Chapter VI we are to see what all this means, as far as living together well is concerned.
[ p. 73 ]
FIRE AND THE METALS AS PHASES OF MAN’S HARNESSING OF NATURE
A. Man’s Conquest of Fire
B. Man’s Conquest of the Metals
Questions to Keep in Mind while Reading This Chapter
- Just what does it mean for man to multiply his powers as compared with merely adding to them?
- What have been the results of man’s conquest of fire?
- What have been the results of man’s ability to command the metals?
- How much better is it for man to be able to use science as his servant?
Man’s progress is a story of his increasing powers. — Our account in Chapters I and II of man in early groups or societies shows that by the time of neolithic man our race had seven great powers or abilities.
Three of them were, so to speak, within man himself. The first was his ability to stand erect, which, of course, he had had for many thousands of years. This gave him a command over the use of the levers of his body (after all, our bones are levers of a sort). No other animal possessed this ability, and accordingly man had power to do things no other animal could do. The second was the fact that the thumb was so placed on his hand that he had ability to use his hand as a grasping device. This, few other animals are able to do, and none of them has the thumb as well placed as man. This ability counts for a great deal when it is a matter of making and using tools. It largely explains, too, the shapes and forms of man’s tools. The third was his power of [ p. 74 ] thinking and planning, in which he greatly excels all other animals. This, as we have seen, was partly the result of his having developed the power of speech. Since speech and communication are to be discussed at length in Part III, we shall not spend more time upon them now.
Out of these three great abilities there grew four other abilities. They were, first, his ability to make fire, and we have seen that quite early he learned how’ to make fire at wall; second, his ability to make and use tools, and we saw that even Neanderthal man had crude tools; third, his ability to command animal life through wMat we call the domestication of animals; and fourth, his ability to command plant life through what we call agriculture. The third and fourth stages had been reached by the time of neohthic man. As we go on with our study, we shall see that all of man’s later progress rests on these seven great foundation stones.
Multiplication of powers vastly more important than mere additions. — There is a very interesting fact about the rate of man’s progress. That rate has been most uneven and irregular. For long periods of time he would make very slow progress. Then something would happen or something would be discovered that would let him progress at a rapid rate for some time. This would be followed by another period of slow progress. And so on. We say that in the periods of slow progress man was merely adding to his powera, whereas in the times of rapid progress his powers were being multiplied.
For example, for many thousands of years man’s powers or abilities as a food-getter were weak, and they developed quite slowly. In the main he appropriated what nature, of her own accord, gave him. When he learned to till the soil and to raise foodstuffs, it meant a great increase in his power to live well. It multiplied his ability to get food. But after [ p. 75 ] he had once learned to raise a few plants, additions to his list of plants were mere additions to his powers rather than multipliers of them. So also when man learned to domesticate animals, it greatly increased his power to live well. These domesticated animals gave him a regular food supply, and many of them were important to him as burden carriers. But once man had learned the trick of domesticating animals, any new animals added to his list were mere additions. As we go on in our study, we must notice what tMngs have been multiplications of man’s powers and what things have been mere additions. The multipliers are far more important.
We begin with a study of man’s work as a harnesser of nature.
¶ A. Man’s Conquest of Fire
(How the fire-making and fire-using abilities of neolithic man have been multiplied.)
Fire is one of the great multipliers of man’s powers. — Anyone will quickly become convinced that fire is a multiplier of man’s powers if he thinks of the ways fire has acted upon scores of things he uses every day. It would take dozens of pages to make a complete list of these ways, but here are some samples. Fke is necessary to make all of the bricks, tiles, piaster’, and metals that we use. It furnishes the heat that runs the engines for sawing the wood for our houses and for making our clothing and household furnishings. Glass and pottery cannot be made without it. Houses are heated by it. Electric lights are but one of its forms. It enables us to run our railroads, our street-car systems, and our automobiles. It cooks our food. We use it to increase the usefulness of almost everything we can think of. It is truly a multiplier of our powers.
[ p. 76 ]
The first steps in man’s harnessing of fire were taken in the dim past. — Man was a user or an appropriator of fire long before he could kindle or create it.
“Natural” fire. — In that far, dim past when he did not know how to kindle a fire, he used fires that nature kindled just as she does to-day. In some parts of the world there are volcanoes whose overflowing lava starts fires in woods and other uncleared land. Cases have been known where earthquakes have dislodged stones, which have crashed down a mountain side and have made sparks that set the grasses on fire. Lightning frequently kindles some tree that it strikes and might even kindle vents of natural gas in certain parts of the world. Then, too, cases of what we call spontaneous combustion occur. This has occurred in coal inines by water seeping in and setting up a chemical action which resulted in fire. We have had stories, too, of such fires being started in masses of decaying vegetables or animals. It is said, for example, that fire has been started by the decaying of a whale cast up on the shore. Dry trees sawing against one another in a high wind have started fires. These are samples of ways in which nature kindles fires.
Fire keeping. — No doubt man at first just used these fires of nature where he found them. Gradually, through thousands of years, he learned to carry “natural fire” to places where he needed it and to “keep” it by carefully feeding it fuel, and by “ covering ” it with ashes. Among these primitive peoples, the duty of keeping the fire was a most important one. It does not surprise us in the least to learn that among many peoples the careful keeping of the fire grew to be part of their religion, and continued to*be so even after they had learned how to make fires themselves. Such a religion of course came down to them from the earlier days when the death of the fire was nothing short of a calamity for the group.
[ p. 77 ]
Fire making. — Fortunately for man, he learned how to make this great multiplier, fire, at a very early stage. We know that Neanderthal man had fire, and some scholars believe that he could make his fire by striking iron pyrites together and letting the resulting sparks fall on dry tinder (see page 18). We know that neolithic man had learned to make fire by whirling one dry stick upon another (see page 35).

Fire Keeping among the Romans
Fire keeping became a religious custom. In the temple of Vesta, young girls kept the sacred lire burning day and night. A great many peoples have had religious rites in connection with fire keeping.
We are quite certain, then, that man has known how to make fire for many thousands of years.
Your great-grandfather saw the next multiplication of man’s fire-making ability. — And now we come to a most interesting fact. Neolithic man knew how to make fire by rubbing sticks together, and by the “strike-a-light” method of the pyrites; and man since then has kept on making fire by such simple methods until very recently. For thousands and thousands of years there was almost no change except [ p. 78 ] some improvements of the tools. He learned to use the flint and steel instead of pyrites, but there was no multiplication of his fire-making ability until the match was invented.
When we want a fire to-day, we make it without any trouble at all by using a match. We never stop to think that the match is one of the most wonderful things in the world and that it was many thousands of years before firemakers learned to use one. It was not, indeed, until after the time of George WasMngton that men learned to make fire by matches. In 1805, a chemist in Vienna invented an outfit for fire making which consisted of a bottle of sulphuric acid into which one dipped wooden sphnts tipped with a mixture of sulphur, chlorate of potash, and sugar. These matches cost about five cents each and were very unsatisfactory. They might or might not catch when dipped in the acid, and frequently they would smolder and sputter, throwing the acid about and spoiling both the temper and the clothes of the firemaker.
It was not until 1827 that an English chemist, John Walker by name, tipped a splint with chemicals that would burst into flame when rubbed on sandpaper, and thus made the first friction match. A few years later, in 1833, by the use of phosphorus in the chemicals, a match was invented that could be lighted without the need of carrying sandpaper around. We then had the phosphorus match. Nowadays a hundred matches can be bought for a cent and we use in the United States, so it is said, over one hundred and fifty billion matches a year. This means that each man, woman, and child uses, on the average, four or five matches a day. Just think what this means in ability to cohimand fire at almost any time or place! Fire is now a servant at our beck and call.
We should remember that man became able thus to command fire because he had developed the science of chemistry. [ p. 79 ] His knowledge of chemistry enabled him to harness the forces of nature in such a way that he could make the friction match. All this was such a short time ago that there are people still living who remember when flint and steel and tinder box were used in almost every household.

Even more recently we have harnessed electricity to make heat for us. We use it in great furnaces or retorts in industry, and it is rapidly coming into use in our households for cooking and other purposes. Probably there are few of us who have not seen and used an electric iron. An Iroquois would never have believed that Heno, the Thunderer, would one day be harnessed to make steel, or to iron a handkerchief. But then, an Iroquois was only in the appropriative and adaptive stages of progress. The creative stage was still to come.
Man’s use of fire in keeping warm. — We shall talk a great deal of the use of fire in modern industry when we discuss the making of iron and steel, the development of transportation, and the providing of power to run machines. Let us at this time see something of how we have used heat as a means of keeping ourselves warm.
The open fire. — The first use of fire for this purpose was seen when quite primitive man kept his fire just outside the crude shelter in which he hved. Of course, most of the heat went to waste in any such scheme. Later he built his fire inside his cave, or hut, or wigwam, or long house, or whatever other shelter he used. In some parts of the world where the weather never grows very cold, houses are still heated by open braziers, which are carried from room to room as they are needed. The fumes are not pleasant or healthful.
[ p. 80 ]
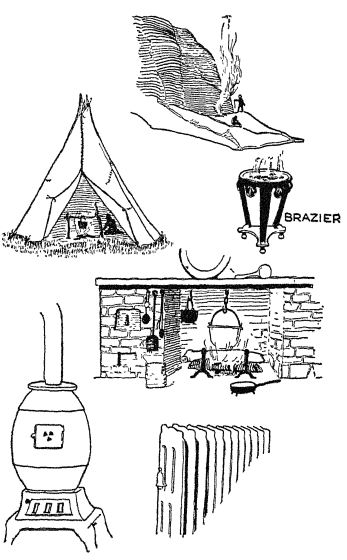
Probably a long time went by before man became wise enough to cut a hole in the roof to let the smoke of his fires out; and no doubt thousands of years went by before he learned to put a couple of poles up by that hole and make a skin windbreak that would cause the smoke to be sucked out more effectively. We are so accustomed to chimneys that we can hardly realize that there were few chimneys in England before about 1200 A.D. And, of course, a chimney is not merely a great convenience in carrying out the smoke. It also creates a draft and makes the fire burn better.
Grates, stoves, and furnaces. — Once the chimney had come into use, houses could be heated by means of the familiar open grate. This also is wastrful of fuel, since eighty-five or ninety per cent of the heat goes up the chimney. A stove invented by our own Benjamin Franklin in 1744 was a great step forward. This stove stood out in the room and radiated [ p. 81 ] heat all around. Although there were stoves earlier, Franklin’s invention brought them into general use and thus enabled people to get from forty to sixty per cent of the heat out into the room. Franklin also made the first attempt at a hot-air furnace. This method began to have general use early in the nineteenth century. So also did the modern hotwater furnace, although we read of the occasional use of hot water to heat houses in ancient Rome.
As we think back over this story of the use of fire for warming ourselves, we are again struck by the fact that for thousands of years progress was quite slow, and that our modern methods are very new. Man is just beginning to have a chance to live well.
Man’s use of fire for light. — Primitive lighting. — The fire of primitive man was, of course, also his light. As time went on, he learned that certain kinds of wood burned more brightly than others, and from these he made rude torches. Then, too, fats when thrown on the fire must very early have given him a hint that they could be used for hght. He gradually learned to use a hollowed-out piece of soapstone, or a clay vessel, to hold the oil or fat, and a sort of rude wick. This was the first lamp.
It is hard to beheve, but for thousands and thousands of years man’s progress in light making was merely a story of additions. This primitive lamp gradually assumed more pleasing shapes; it became a closed vessel with the wick sticking out of a hole in it; it was gradually shaped so that it had several wicks; candles were made (an early form was the candle fish, which was so full of grease that when a wick was thrust in it, it served as a candle) ; lamps were later incased in glass (these were not chimneys) to protect them from the wind. But after all has been said, it was not until about 1800 that man got a better light than the flickering, [ p. 82 ] smoking, and too frequently foul-smelling light of torches, grease lamps, and candles.
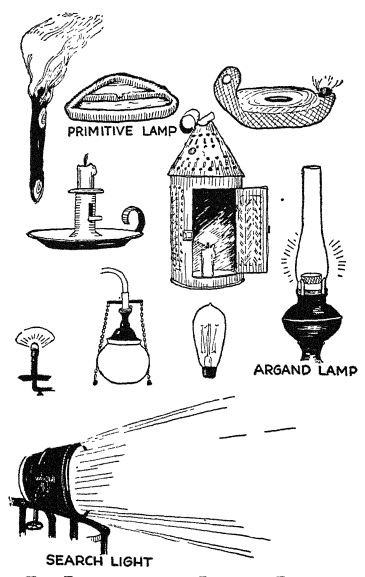
The wonderful lamp chimney! — This improvement also was connected with the development of the science of chemistry. About 1784 a Swiss chemist, Argand, discovered that he could have a circular wick instead of a flat wick and could arrange to have a draft of air alongside the flame. The oxygen in the air caused a brighter flame with much less smoke. Quite by accident Argand’s younger brother broke a bottle over one of these flames in such a way that it acted as a chimney, and he noticed that the flame was very much better. This was the first glass chimney, and it multiplied the lighting power of the lamp. Think what this meant to those who would read or work by night!
New fuels. — Improvements in the fuel used for lighting are also quite recent. Animal fats and whale oil were the [ p. 83 ] usual fuels until within the last one hundred years, although even the ancients knew of natural gas and knew that it burned with a fairly bright flame. People did not learn how to manufacture gas from coal until in the eighteenth century. In 1739 an Englishman by the name of Clayton described how gas could be produced from coal by heating the coal in a closed vessel, or retort, but it was not until 1792 that one Murdock lighted his house and offlce by gas. In the period from 1810 to 1830 gas began to come into use for lighting streets, supplanting the old whale-oil lights. This, you will notice, was only a hundred years ago, and in the older streets of both this country and England there still are houses with devices on the railings with which to extinguish the torches used in lighting guests to carriages. Kerosene did not become available until about 1850.
Electricity. — Of course, electric lighting is still more recent than lighting by gas. It was not until 1862 that an electric arc lamp was put into regular service. There were only two exhibits of electric hghting — and they were regarded as great curiosities — at the Centennial Exposition, which was held at Philadelphia in 1876. Edison first exhibited his incandescent lamp at his laboratory in Menlo Park, New Jersey, in 1879. It was not until 1882 that the first central power station for making electricity was constructed.
We are so accustomed to stepping outside the house at night into a street hghted by electricity that it is hard for us to understand how recently man has harnessed this natural force. We need to remember that people are still hving who walked on streets that must have looked very much as our streets look on those rare nights when something has gone wrong at the power plant and the town is almost in darkness. And as for our ability to press a button and have a flood of light in our home, or to strike a match and hght the gas, or, [ p. 84 ] for that matter, to strike a match and light a kerosene lamp, these things are events in the lifetime of our own father or grandfather.
A chart shows the newness of our control over fire and light. — As a means of seeing how very recent our present ability to command fire and light is, let us go back to the chart we made in Chapter I. The chart in that chapter (see page 6) was one covering the length of time man has been a good communicating animal. If we were to try to locate on that chart the more important multiplications of man’s heating and lighting power, we should find them all crowded at the extreme right end of the line. Indeed, they would be so crowded together that they would not show well on the chart.
Let us, then, make another chart showing only the last few centuries of man’s progress in fire making, and let us remember that even these few centuries will take in the more important happenings since the time of neolithic man.
Make on the sheet of note paper you are using for your charts this heading:
Six Hundred Years of Man’s Harnessing of Fire and Light

Place the date 1900 over at the right side of the paper and draw a line out to the left, letting each one hundred years be represented by a space one and one-fourth inches long. Then every one eighth of an inch will stand for ten years. Carry the line back to 1300 a.di Locate on this line the more important devices that man has developed in the field of heating and lighting as far as we have discussed the matter. Do you find that they are very recent?
[ p. 85 ]
¶ B. Man’s Conquest of the Metals
(How the metals have multiplied our powers : how we have secured plentiful iron and steel.)
The multiplier, fire, has given man another great multipher, the metals; for fire has enabled man to take certain kinds of stones or rocks called ore and melt out useful metals. It is true that small amounts of a few metals, such as gold and copper, are occasionally found free from the ore. However, so little metal can be obtained in this free state that it is fair to think of fire as the great metal-giver of man.
How metals multiplied the powers of early man. — We can see how the metals multiplied the powers of early man by studying their effects upon his tools.
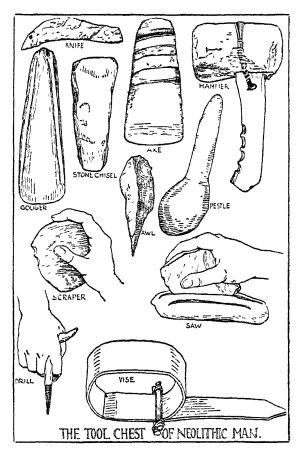
Neolithic man had many tools. — If we think back over the account of neolithic man, as illustrated by the Iroquois, we remember that he had a really surprising collection of tools. He had simple and rude samples of all the important tools we use to-day. Of what we call cutting tools, he had knives and gougers, which he worked by pressure; axes and stone chisels, which he worked by blows or shock; and saws made of flints with rough edges, or of sharks’ teeth or other pointed teeth set [ p. 86 ] in wood. Of what we call smoothing tools he had scrapers, smoothers, polishers, and grindstones. You will remember how the Iroquois women scraped the deer skins which they were curing, and how the bows and arrows were smoothed and polished. Of what we call fracturing, crushing, and grinding tools, he had hammers, pestles, and grinding apparatus such as those he used in grinding grain. Of what we call perforating tools he had needles, prickers, awls, punches, and drills of various sorts. We have seen uses he made of all of these perforating tools with the possible exception of his drills. We know of his drills, however, for the same tools that were whirled to make fire were pointed with a bone or thorn and whirled to make a hole. Of what we call grasping or joining tools, he had tongs, crude vises, lashings, and glues. Altogether he had quite an assortment of tools.
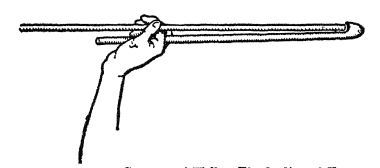
A Throwing-stick
This shows the Australian natives’ method of using a throwmg-stick or spear-thrower The throwing-stick was really a sort of lever enabling them to hurl the spear greater distances
Defects in his tools. — These tools of neolithic man had two great defects. One was that he did not know how to apply much power to them. It is true that he had learned to “haft” them, or put handles on them. It is true, also, that in such devices as the throwing-stick or the bow and arrow, he had the beginnings of what we call to-day the use of power. But he had the merest beginnings.
The other defect of primitive tools was that they were made of very unsatisfactory material. Most of them were of stone, though sometimes wood was used, as were also bone, [ p. 87 ] horn, and even ivory in places where ivory could be secured. Now, all these materials are quite brittle and very likely to break when put to hard use. Imagine the difficulties you would have if all your knives, chisels, scissors, needles, and hammers, were made of such materials. You would surely feel that you had very unsatisfactory tools. You would surely feel that metal tools would be much better and would greatly multiply your powers.

This man is plowing, harrowing, pulverizing, and seeding in one operation. Neolithic man, lacking metals, could not make such giant machinery. He could not command nature as well as we do to-day in agriculture. See page 33 for a picture of his agricultural tools.

A Few Bronze Instruments
These bronze-age peoples could live much better than neolithic peoples because they had metal sickles, awls, fishhooks, knives, saws, spoons, and the like. Compare this picture with the one on page 22.
Metals, especially the alloy bronze, removed one defect. — Nobody knows when metals first began to be used, but even before this communicating animal, man, had learned to write, he had learned to use metals. Nobody knows what tribe or race of man used metals first; nobody knows what [ p. 88 ] the first metals to be used were. Probably, different metals were used first by different peoples, depending, of course, upon the metals with which nature had provided their particular territory. Some writers think that the very first metals to be used by man were copper and gold, because these metals are found in many places free from ore and their glitter would attract the attention of primitive man. If this is true, primitive man may have thought of them as very interesting kinds of stone which he could appropriate and adapt into useful and pleasing shapes.
As time went on, certain peoples who lived where there were both copper and tin made the discovery, very likely by accident, that when these metals were melted together in a fire, the result was a new metal, bronze, which was for many purposes very much better than either the copper or the tin. This bronze is a so-called alloy. Alloys are very interesting. They are quite likely to have [ p. 89 ] qualities not possessed by either of the parent metals. This is true in the case of bronze. It is harder than either tin or copper, can more readily be cast into satisfactory form, and can be given a better and more durable cutting edge.
The discovery of bronze multiplied man’s powers. All his older and unsatisfactory stone or bone tools could now be made of bronze. These metal tools gave him greater command over nature in his tree-cutting, boat-making, and soildigging. Once this multiplication of powers had taken place there came next the slow process of addition, and man’s tools steadily grew more numerous and better.
All other metals have yielded to iron. — But copper and tin are, after all, not very plentiful. In parts of the earth neither exists; in most parts of the earth they do not exist together and they must be together before bronze can be made. The one metal that can be found in almost every part of the world, although usually in the form of ore, is iron.
It is not surprising that it took man a long time to learn of iron. The ore in which it is found does not look very different from any other stone, so far as a savage can tell. Quite likely early man found a few pieces of iron in the “free” state. But it must have been by some accident that man first learned to make it. It may be that iron metal was first seen in the embers of some camp fire as a result of a chunk of ore being placed in the fire. There may be truth in the old tradition that in one part of the world iron was discovered as the result of a great forest fire which burned over an area in which there was iron ore. No one to-day can be sure when, where, or how iron was first made. We only know that man has had it a very long time, and that there were some parts of the world in which he had it before he had bronze. Suppose we say that he has had iron about as long as he has been able to write.
[ p. 90 ]
Primitive methods of iron making have continued down to the last four himdred years. — Although we do not know when, where, or how iron was first made, we do know the main steps that man took in mastering this metal, which is now the most important single commodity he uses, with the exception of air and water.

Primitive Iron Forging
One of these savages is working a crude bellows; the other is ready to drop small lumps of charcoal and iron ore into the fire (which has not yet been built) and to hammer the iron with stone tools. By working hard they can produce ten pounds of iron in a day. Compare this picture with the ones on page 91 and page 93.

The Catalan Forge
Compare this picture with the early savage forge shown on page 90 and with the modern blast furnace shown on page 94.
Early iron production. — As you would expect, his first steps in mastering iron were very feeble ones. We can see something of what they must have been by noticing how, within the memory of living man, savage people in Africa made iron. It is a fair guess that man of long ago made iron in something the same way. The picture shows two naked savages squatting on opposite sides of a primitive forge. One of them is working a crude bellows which sends a blast of air [ p. 91 ] through an underground tube to the place where the fire for reducing the ore is to be built. The other savage is ready to keep dropping small lumps of iron ore and charcoal into the blaze. These two savages, working, pumping, sweating all day long might have at the end of the day a lump of iron which would weigh as much as ten pounds. You can see that they are ready to hammer this iron with stone tools. By hammering, reheating, and hammering again and again they could get most of the impurities out and have very good iron.
The Catalan forge. — Astonishing as it may seem, this primitive way of making iron, with only some additions, continued to be the way that iron was made down to about the time of the discovery of America by Christopher Columbus. We can neglect these minor additions and come at once to the Catalan forge, so called because it was developed at Catalonia, Spain. This was the best device for making iron that man had until about four hundred years ago. Such furnaces have, indeed, been used in our own country within the memory of your grandfather.
Above is a picture of the Catalan forge. At the bottom of the chamber was put charcoal; the fire was started and blown by [ p. 92 ] a current of air from the bellows. Above the fire were piled alternate layers of iron ore and charcoal. As the charcoal burned away and as the iron ore shrank in size in the process of forming iron, more ore and charcoal were placed on the top of the heap or “charge,” and this was continued for hour after hour.
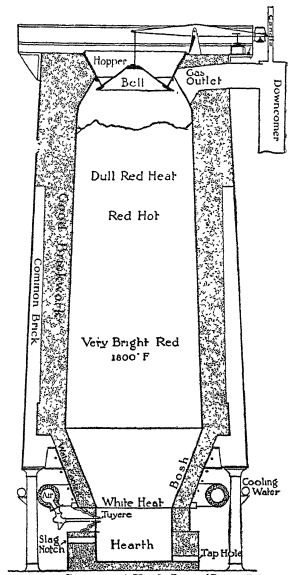
Diagram of a Blast Furnace
Today such a blast furnace may be one hundred feet high. At the bottom are the tap holes for the iron and for the slag. Just above the slag hole is the tuyhre where the blast of hot air (we now heat the air used in the blast) is thrown into the furnace. The “charge” of limestone, ore, and cohe, which is to-day used instead of charcoal, is at white heat down near the bottom, red heat a little higher up, a dull red heat near the top. A great volume of hot gas fumes pours out the gas outlet at the top. These gases are made use of as a byproduct, The charge is thrown in at the topthrough an adjustable hopper and bell. The bell closes the opemng after the charge has been put in.
When the blaze was stopped, there was found in the bottom of the furnace a ball or spongy mass of iron (it was called a bloom and the furnace was called a bloomery), still mixed with various impurities. This spongy mass was taken out and hammered and reheated and hammered and reheated and hammered again and again until most of the impurities had been removed. The result was a very good quality of metal, as good, indeed, as we are able to make to-day. Such a furnace, wasteful as it was, could in a day’s time do the work of two hundred of the sweating savages shown in the picture on page 90. Man had made that much progress in his mastery of iron.
Iron scarce in those days. — But such methods did not make it possible to produce very large quantities of iron. It has been estimated that in the year 1600 the amount of iron made by the whole world was so small that if it had been divided up evenly among the inhabitants of the world a tablespoon or, at the largest, a teacup would have been an appropriate measuring device. Since iron was so scarce, it was expensive and had to be used very carefully. W”e can get some idea how precious it was from an account of the way they used it in medieval England. It was sold by the “piece,” each piece weighing only about four poimds. A little faggot of these bars would be carefully treasured in each manor, and handed out bit by bit to make such things as plowshares, nails, and horseshoes. No one would think of being so extravagant as to use iron to make teeth for harrows, or to put tires on [ p. 93 ] wheels. It had to be saved for more important uses.
The blast furnace and appropriate fuel enormously increased our command over iron. — The next great surge forward in man’s mastery of iron was what we call the blast furnace. This blast furnace is just an improvement of the old Catalan forge, but it is such a great improvement as to be a multiplier of man’s powers. As men went on using the Catalan forge, they found ways to make the air blast much stronger. They could, accordingly, make the height of the furnace greater and still force the blast of air up through the charge. The higher furnace and the better blast of air made possible a very much hotter fire. This hot fire melted the iron which was in the ore to a liquid (the old Catalan forge was not able to do this) afid this liquid would trickle down to the bottom of the furnace.
[ p. 94 ]

They found, too, that by mixing limestone with the charcoal and iron ore, many of the impurities of the ore would, in the fierce heat, combine A\ith the limestone and also flow to the bottom of the furnace as another liquid. The liquid iron was heavier than the hquid impurities or “slag,” so that there would be two layers of liquid at the bottom of the furnace. From time to time, the upper layer, slag, could be drawn off through an opening. The hquid iron could be drawn off through a lower opening. Since the iron and the slag could both be drawn off without stopping operations, men could keep on reducing the ore day after day, and week after week. As fast as the limestone and charcoal and iron ore turned to iron and slag, added material could be thrown into the top of the furnace without stopping the work at all.
It is said that the first blast furnace was made in Belgium about 1340, but this new device was not used very widely until about 1550. For that matter, mot until man had learned to use anthracite coal and coke, in about 1750, did the blast furnace really come into its own. Anthracite coal and coke (coke is made of soft coal) are both firm enough to bear the [ p. 95 ] weight of great masses of iron ore and limestone without mashing down. This makes it possible to use great furnaces 80 to 100 feet high and still force the air up through the charge. Such a blast furnace will turn out as much iron as could be made by 200,000 of the toihng savages shown on page 90.
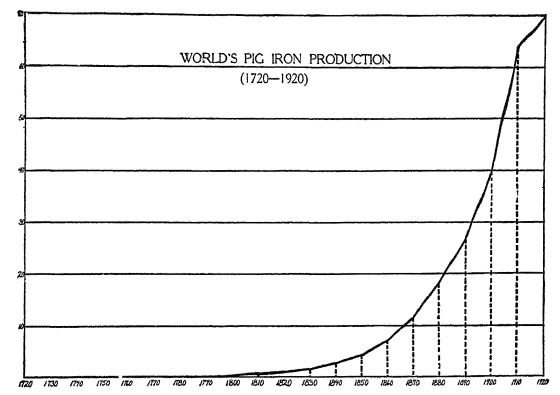
The top line stands for 70,000,000 tons of pig iron The amount made annually before 1800 hardly shows on the chart. It is clear enough that the large output of iron is a matter of the last seventy-live years.
As a result iron is plentiful. — The chart shows that it is only since man has learned to use the anthracite or coke blast furnace that he has become an enormous producer of iron. Indeed, it is only in the last hundred years that the amount of iron produced dach year makes much of a showing on the chart. The same facts may be told another way. If all the iron made in the world in 1500 were put in a wall eight feet wide and six feet high, the wall would reach for seven [ p. 96 ] city blocks. If the iron that is made in a single year to-day were put in a wall of the same size, that wall would reach from Chicago to New York and three hundred miles out into the ocean. If we let the heavy black line shown below represent the amount of iron made in a single year to-day, the little speck at the lower left represents the amount made in 1500. Iron does not need to be doled out to-day in little fourpound bars! We use it lavishly, and its lavish use is one of the reasons why we are able to live better and more comfortably. Just take a look around and see the quantities of metal (mostly iron) which we use to-day! What kind’ of life would we be living if we had to do without it?
The World’s Production of Iron in 1920 and 1500 [1]
The big line shows the iron produced in 1920. The little speck at the lower left shows how much was produced m 1500. As was shown in the preceding chart, the great increase in production IS quite recent.
Man uses chemistry to make steel cheaply. — Even yet the full story of man’s control of his servant, iron, has not been told. Useful as iron is, steel is much better, for with the same weight, steel has much greater strength and hardness. Now, ordinary steel is just iron with a small and proper percentage of carbon in it. Once man had learned to make iron cheaply and in great quantities, the next problem was to free this iron from its impurities and then be able to mix with it just the right amount of carbon or other material to make the kind of steel desired.
Early steel making. — As we haVe seen, the earliest steelmakers burned and hammered the impurities out of the iron; they mixed carbon with it by heating it with charcoal, which is mainly carbon. Some of these early steelmakers became [ p. 97 ] very skillful, and made as good steel as we make to-day. The trouble was that they did not know chemistry and did not know how to measure the different elements as does the chemist to-day. In consequence, only a few highly skilled and intelhgent people developed the “knack” of making good steel. They “judged” when conditions were just right. But when chemical knowledge became available, man no longer needed to rely on guesswork or judgment. The chemist is able to measure accurately. Furthermore, in addition to giving us exact measurement instead of “judgment,” he is able to tell us many new ways of removing impurities.
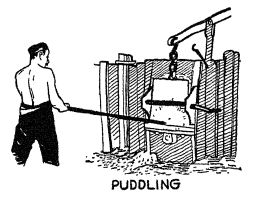
Let us see some of the more important steps by which man has become able to make good steel in great quantities.
Cort’s puddling process. — The liquid iron that flows from the blast furnace has many impurities in it. A great step in purif3nng this iron was taken when Cort invented in 1784 his puddhng and rolling process. He built a huge furnace, the bed of which was made of certain material that would act in a chemical way on the hquid iron poured into it. After the hquid iron had been poured in, he caused an intensely hot flame to pass over its surface while it was being continuously stirred or puddled by a workman. Graduahy, as a result of the chemical action of the bed and of the flames, most of the impurities either burned out of the iron, or trickled off as slag. The result was a spongy mass. Cort then took this from the furnace and ran it backward and forward between enormous grooved rollers that squeezed stiH further impurities out of the metal. This is quite hke what the black savages did when they reheated and hammered [ p. 98 ] and reheated and hammered the spongy metal. The difference is that in Cort’s day man had large furnaces and machinery to do the work for him and had learned to call chemistry to his aid. This is, of course, a very great difference. It means that iron can be purified much more cheaply. After it is purified, carbon can be introduced and then we have steel.
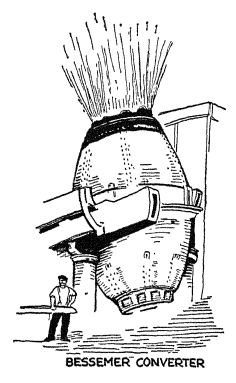
The Bessemer process. — The next gi’eat step was taken when Kelly in America and Bessemer in England invented (1846 to 1858) a device for removing impurities from the liquid iron in from ten to twenty minutes, as compared with the hours needed for the Cort process. The Bessemer process sounds so simple that it may seem surprising that it was not discovered sooner, but, after all, we learn very slowly. All that Bessemer did (and even this had been done by Kelly earlier) was to put the liquid iron into an enormous vessel, the sides and bottom of which were lined with the right kind of chemical material, and then to force small streams of air up through the molten mass. The oxygen in the air greatly increased the temperature of the liquid metal and burned out the impurities. The great flame coming from the top of the vessel (with its colors changing as the various impurities disappear and so cease to color the flame) is a wonderful sight.
The open-hearth process. — About the same time, 1856 to be exact, a man by the name of Siemens found still another way to remove the impurities from the liquid iron. He developed what is called the open-hearth process for making [ p. 99 ] steel. This process is not greatly different from the principle used by Cort in his furnace. The hot liquid metal is placed in a furnace and an intensely hot flame, much hotter than could be developed in Cort’s time, plays over the liquid metal. This flame, together with chemical action caused by materials in the bed of the furnace, slowly removes the impurities without the necessity of using the puddling process of Cort.
The end sought in all these modern processes is that of getting the iron in a fairly pure condition. Once this end has been gained, it is, of course, possible to introduce carbon or other materials in such quantities as may be desired, and then we have steel. The work is done according to rules carefully worked out by chemists. This precisely controlled, scientific making of steel is a far cry from the steel making of our two naked Africans.
Electricity and alloys. — To-day the making of steel is in a new stage. For one thing, electricity is now being used as the heating device. This makes possible a far more intense heat and at the same time gives a far more delicate control of the heat than we have ever had before. The result is an even finer product. For another thing, we have carried our knowledge of chemistry to the point where it enables us to make wonderful steel alloys, such as manganese steel, chrome, tungsten steel, and nickel steel. We should expect these alloys to be wonderful things because, as we saw in the case of bronze, (page 88) an alloy is hkely to have properties and powers not possessed by the parent metals. Nickel steel will serve to illustrate them all. The combination of nickel and steel gives us a metal that is exceedingly hard and that at the same time does not crack easily. For this reason it serves admirably as armor on our war vessels, as shafting on our machinery, and in general as good material for all uses which demand great power of resistance.
[ p. 100 ]
Cheap iron and steel have meant a different world to live in. — All these great improvements in making iron and steel have made them so much cheaper that we no longer have to be economical in using them. The result is that we make such great use of steel that many writers call this the “age of steel.” The name is a good one, for steel to-day enters into almost everything we do.[2]
The age of power. — For one thing, this age of steel has made possible an age of power. When one thinks of our power makers to-day, he thinks of steel grates, steel boilers, steel cylinders, steel pistons, steel driving shafts in our engines. He thinks of dynamos, tractors, automobiles, and cranes, and these are all made mainly of steel. We could not have harnessed power if we had not first harnessed steel.
The age of machines. — For another thing, this age of steel has made possible our age of machines. As long as man had to depend upon wood or even upon iron to make his machines, these machines would not stand much strain. They were too weak for the driving powers man had harnessed. But when this giant, steel, was harnessed, we had material out of which we could make our modern agricultural machinery, our printing presses, our textile machinery, our shoe-making machinery, our war machinery, and all the other clattering servants which man drives to do his will. This machinery gives us goods of better quality and greater quantity than we could have had without it.
The age of transport. — Then, too, this age of steel has made possible an age of transport. As long as we could use merely wood or iron, rails could not be made which would carry heavy loads; bridges could dot be built which would [ p. 101 ] meet the strain of heavy traffic; wheels could not be made to carry heavy burdens. With an abundance of steel all this was changed. Steel rails, steel bridges, steel wheels, steel locomotives, and steel sheets made possible the use of steel freight cars, steel passenger coaches, and steel ships. The result has been an abihty to carry goods cheaply in great quantities. We may thank steel for the fact that the total cost involved in moving the hide to the tannery, the leather to the shoe manufacturer, and the shoe to the consumer is only a few cents per pair. And similar statements could be made of most of the goods we use to-day.
Our dependence upon iron. — Even this does not tell the full story of the effects of the age of steel upon our living together. Its furnaces, its steel beams, its stoves, its piping, have made our homes very different from what they would be without its presence. Its use in our hospitals, in our water works, in our sewers, and even in our medicines (for iron is one of our most important medicines) has meant a great deal in the realm of health. Its automobiles, its Ferris wheels, its roller coasters, its moving-picture machines, its playgroimd apparatus, its phonographs, have a great part in our recreation. The way iron serves us in our food getting is typical. Our bread and meats are borne to us over the iron way from the grain fields and pastures of the West. The iron horse brings us oranges from Florida and California; the iron ship brings us bananas from the Caribbean Sea. Iron machines prepare our canned foods. The cans are made by machines; they are filled by machines, sealed by machines, soldered by machines — everywhere machines; everywhere iron and steel.
Our dependence upon iron is so great that if we were suddenly deprived of it we would drop back almost to the living conditions of Jacob and Esau. Food would no longer be [ p. 102 ] easy to get, nor coal, nor wood, nor wool, nor cotton. Famine would wipe out hordes of us as an eraser removes chalk marks from the blackboard, and where 100,000,000 persons are now there could exist not more than 5,000,000. And all that has been said on the last two pages is but the merest beginning of a catalogue of the ways in which cheap iron and steel have made this a different world to live in.
Abundance of iron and steel is recent. — Let us now review our discussion of man’s mastery of metals, as illustrated by iron, in such a way as again to make it clear to us how recently man has become able to live well. On page 84 we had a chart showing six hundred years of man’s harnessing of fire and light. Let us now make a similar chart, giving it this heading:
¶ Six Hundred Years of Man’s Harnessing of Iron
As we insert on this chart the important events in the harnessing of iron and steel, it will become very clear that we have only recently secured our large command over metals. Such a thought makes us wonder what the future has in store for us. Man is just beginning to have a chance to live well.
¶ Problems
- Define or explain
Multiplication of powers, Alloys
Chemistry, Catalan forge
The flint and steel, Coke
Bessemer process, Slag
Open-hearth process, Argand burner - What things can you mention tha’fc man is better able to do because of his ability to stand erect? Have you ever seen animals “rear up” so as to free their front legs for action?
- What things can you mention that man is better able to do because of his “free” thumb? Can you name any other animals that make [ p. 103 ] similar uses of their forefeet? If you can, are they as skillful as man at it?
- Name as many things as you can that show man’s power to live well was greatly increased when he learned to command plant life through agriculture. Did it mean more food? Better food? More dependable food? Does it mean the same things to-day as far as food is concerned? Does it mean anything today as regards clothing? Shelter?
- Show in detail that man’s power to live well was greatly increased when he learned to command animal life through domestication of animals.
- One writer says that man has not added any new member to his list of domestic animals in the last 5000 years. Do you know whether he has done much in that period by "way of making improvements in the animals he had already domesticated? Why do we teach animal husbandry” at our agricultural schools to-day? Is man harnessing nature when he improves the breed of his animals?
- Can you name a single thing you have used to-day that has not been to some extent affected by man’s use of fire? Do you suppose that in your high-school and college work there will be much studying of heat and its consequences?
- Explain why early peoples should think it so important to have good fire keepers that they made fire keeping the special duty of certain members of the group.
- Draw up a list of reasons why the match is better than the flint and steel. Just how did the chemist harness nature when he gave us the match?
- Think back over the history of man’s use of fire. Point out an appropriative stage; an adaptive stage; a creative stage. In which stage has man greatest command over fire?
- Think back over the history of man’s use of fire. What events are so important that it is fair to call them ‘"multipliers”? Is the harnessing of electricity important enough to be so called?
- Think back over the history of man’s use of light. What “multipliers” can you mention? Did science give us any of them?
- Can you name a single thing you have used to-day that has not been to some extent affected by man’s use of metals? Do you suppose that in your high-school and college work there will be much studying of metals and their consequences?
- Think back over the history of man’s use of iron. What events are so important that it is fair to call them “multipliers”? Was the discovery of how to make coke as important as that? Was Bessemer’s invention? [ p. 104 ]
- In the history of man’s use of metals find an illustration of the appropriative stage; the adaptive stage; the creative stage. Were the two African savages in the creative stage?
- Some writers, speaking of stages in man’s progress, talk of “The bronze age.” Why could man live better in “the bronze age” than in “the stone age”?
- Cite as many instances as you can ivhere the knowledge of chemistry is used in modern making of iron. Was chemistry at ivork when the African savages made iron, even if they were not aware of it?
- Look at the chart on page 95. Write down a list of the thoughts which it brings up in your mind.
- Draw up in parallel columns a comparison of steel making by (1) the African savages and (2) modern methods. Here is a start; carry it on as far as you can.
| Points of Comparison | Savage Methods | Modern Methods |
|---|---|---|
| 1. Use of science. | 1. Not aware of science | 1. Much dependence upon science. |
| 2. Amount made. | 2. Small; 10 lbs. per day. | 2. Great; 1 blast furnace equals 200,000 savages. |
| 3. Quality. | 3. Good, but irregular. | 3. Good, and very uni form. |
| 4. Mechanical devices. | ||
| 5. How carbon was put in. |
- Draw up in parallel columns a comparison of the way iron and steel W’ere used in 1500 and are used to-day. Carry on this start.
| Points of Comparison | Use of Iron and Steel in 1500 | Use of Iron and Steel To-day |
|---|---|---|
| 1. Power devices. | 1. Almost none. | 1. Makes possible our mechanical power devices. |
| 2. Machines and tools. | 2. Very little. | 2. The main element in tools and machines. |
| 3. In food production. | ||
| 4. In clothing making. | ||
| 5. In amusements. | ||
| 6. In transportation. |
- “Man is just beginning to have a chance to live well.” Explain as far as light and fire are concerned. Explain as far as metals are concerned. [ p. 105 ]
- Make a list of cases where fire has been harmful to our living together well. Do the same for iron and steel. What is the remedy? Can we as separate persons do anything to help? Can society (in the form of the city or state) do anything to help?
- “It is not enough for man to master nature. He must also learn to be a wise master.” What does this mean?
- Write a theme on the kind of life that would be possible to-day if we had no fire.
- Write a theme on the kind of life that would be possible to-day if we had no metals.
- Make the charts called for on pages 84 and 102.
- Answer the questions at the beginning of this chapter, page 73.
¶ Interesting Reading
Marshall: Readings in the Story of Human Progress, Chapter III.
- How Early Man Tamed Fire (illustrations of early fire making).
- The Manufacture and Use of Artificial Gas (one of man’s invisible servants) .
- Earthenware and Porcelain (fire gives us pottery: our use of ceramic products).
- Harnessing Rays of Light (what the lens has meant for living well).
See also:
Chapter VI, 2. Petroleum and Its Uses (what one natural resource means to us: the need of conservation).
Chapter XV, 4. Lighting a City (how a city performs one task) .
Problems to think over are given in these reading selections.
| II. The Greater Powers of Neolithic Man: the Benefits of Tools, Communication, and Social Organization | Title page | IV. Power and the Machine as Phases of Man’S Harnessing of Nature |