Public domain
[p. 11]
The mantle rock. The great body of the lithosphere is probably composed of solid rock, but the solid rock is very generally covered by a layer of loose material consisting of soil, clay, sand, gravel, and broken rock, known collectively as mantle rock. The mantle rock of many places consists of the disintegrated products of underlying rock formations. The upper part of such mantle rock is constantly being blown away by wind and washed away by water, but it is being constantly renewed by the decay of the rock below. The mantle rock of some other areas, as the northern part of North America and the northwestern part of Europe, consists chiefly of an irregular sheet of commingled clay, sand, gravel, and bowlders, known as drift. The drift was deposited by great glaciers, comparable [p. 12] to those which now cover Greenland and Antarctica. In still other places, especially along the flood plains of streams, the mantle rock consists of deposits made by streams. Along the shores of lakes and seas, there are beach gravels and sands. The thickness of the mantle rock varies from a few inches to hundreds of feet (Fig. 1).

The solid rock. Mantle rock is absent in some places, and there the surface of solid rock appears. It is common on the slopes of steep-sided valleys and mountains, and on the slopes of cliffs which face seas or lakes, and is frequently seen in the channels of swift streams, especially where there are falls or rapids. In all lands inhabited by civilized peoples, there are numerous wells and other excavations ranging from a few feet to several hundred feet in depth, and occasional wells and mine-shafts go much deeper. Even in the shallower excavations solid rock is often encountered, and in most regions excavations as much as two or three hundred feet deep reach it. It may, therefore, be accepted as a fact that the upper surface of the solid rock is nowhere far below the surface.
Varieties of solid rock. If the mantle rock were stripped from the land, the rock beneath would be found to be made up of many kinds of rocks, all of which may be grouped into three great classes. By far the larger part of the land surface would be of rock arranged in layers (stratified rock), and the remainder would be of rock which is without distinct stratification. The rocks without distinct stratification are divided into two great groups, igneous rocks, and metamorphic rocks.
The essential feature of stratified rock is that it is arranged in layers. The layers may be distinct or indistinct, and they may be thick or thin. Thick layers are often made up of many thinner ones.Fig. 2 shows rock which is distinctly stratified.
In composition, much stratified rock corresponds somewhat closely with the sediments now being carried from the land and deposited in the sea; that is, these rocks are made up of pebbles, sand grains, or particles of mud, cemented together. The bedded arrangement of stratified rocks and of recent sediments is the same, and the markings on the surfaces of the layers, such as ripplemarks, rill-marks, wave-marks, etc., are identical. Furthermore, [p. 13] the stratified rocks of the land, like the recent sediments of the sea, frequently contain the shells and skeletons of animals, and sometimes the impressions of plants. Most of the relics of life found in the stratified rocks belonged to animals or plants which lived in salt water. Because of their structure, their composition, their distinctive markings, and the remains of life which they contain, it is confidently inferred that most of the stratified rocks which lie beneath the mantle rock of the land were originally laid down in beds beneath the sea, and that the familiar processes of the present time furnish the key to their origin.

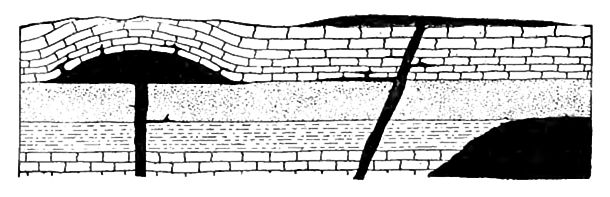
Igneous rocks may be loosely defined as hardened lavas. They sustain various relations to the stratified rocks, as illustrated by [p. 14]Fig. 3, where some of the igneous rock, indicated by black, is represented as lying beneath the stratified rock, and some above it, while some is interbedded with the stratified rock, and some cuts across its layers. From these relations it is possible to tell something of the order in which the rocks were formed. Where the stratified rocks are broken through by lavas, it is clear that the stratified rocks were formed first, and the lavas intruded later. Lava sheets intruded between beds of stratified rock can be told from those which flowed out on the surface and were subsequent ly buried, for in the former case the sedimentary rocks, both above and below the igneous rock, were affected by the heat of the laVa, while in the latter case only those below were so affected.

Most metamorphic rock has cleavage, that is, a tendency to break in one direction rather than in another. The cleavage of metamorphic rock may look much like stratification, but it is really very different. The tendency to break along certain planes is not due to the fact that the rock was deposited in layers originally, as in the case of stratified rock, but is the result of the changes which [p. 15] the rock has undergone since it was formed. The structure shown in Fig. 4 is known as schistosity — a structure characteristic of metamorphic rock. The planes of cleavage, often irregular, are independent of the original planes of stratification in those cases where the rock was originally stratified. Metamorphic rock maybe derived from igneous rock, as well as from sedimentary.
More commonly than otherwise, metamorphic rocks lie beneath sedimentary beds, or come to the surface from beneath them, and they are often broken through by igneous rocks.
¶ Igneous Rocks
How they come to be at the surface. Some of the igneous rocks which appear at the surface and beneath the mantle rock, have been extruded from volcanoes. From some volcanoes, such as those of Hawaii, lava flows in streams, and as it cools, hardens into solid rock. From others, such as Vesuvius, fragments of hot rock are hurled high into the air and fall about the vents. Fragment al material ejected from volcanoes forms pyroclastic (firefragmented) rock. Pyroclastic rock is, in many cases, lava blown into small pieces which harden separately.
Lava sometimes flows out on the surface through long fissures, which extend down to great depths. Such extrusions of lava are called fissure eruptions. There have been few great fissure eruptions in historic time, but in recent geologic time, there were great eruptions of this sort in the northwestern part of the United States, as well as in some other parts of the earth. All rock matter extruded from volcanoes and fissures is extrusive rock.
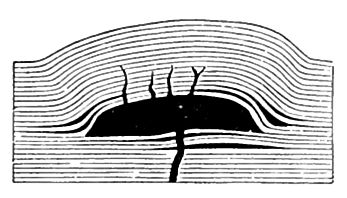
Besides being thrown out or poured out from volcanoes and fissures, igneous rocks appear at the surface in other ways. Much lava which rises from the deep interior does not reach the surface, but is intruded into rock which lies above the source from which it started. If intruded lava fills cracks in the earth, it forms dikes. If it forces itself in between layers of rock, it may take the form of intruded sheets, or sills. If it accumulates below the surface in masses, arching up the overlying beds of rock, it forms laccoliths (Fig. 5). If it breaks and lifts its cover, instead of arching it up, the lava is called a bysmalith. Laccoliths and bysmaliths may [p. 16] be large enough to form good-sized mountains. The Henry Mountains of Utah are laccoliths. Very great intrusions of rock of massive form are sometimes called batholiths. Rock solidified from lava well below the surface is called plutonic rock.
Sills, laccoliths, bysmaliths, batholiths, etc., are formed beneath the surface; but if the covering rock is worn away, as it may be by erosion, the igneous rock appears at the surface. Much of the igneous rock which is now at the surface or concealed by the mantle rock only, is intrusive rock, laid bare by the removal of its original cover.

The larger structural features of igneous rock. Igneous rocks have certain structural features which distinguish them from other rocks. Thus laccoliths, bysmaliths, and batholiths arc massive. [p. 17] This term means not simply that the rock occurs in large bodies, but that the rock has no distinct cleavage. It is not in beds, and it is not schistose. Sills and extrusive flows of lava often take on the form of sheets. When one extrusive sheet of lava overlies another, the succession of sheets has some resemblance to stratified rock; but the materials of the several sheets show little indication of arrangement in layers. The material of the sheets, however, sometimes has a peculiar structure developed by the flow of the lava after it had become stiff from cooling. This structure is known as flow structure (Fig. 6). In some places, the lava, on cooling, develops a columnar structure (Fig. 7) which appears to be the result of contraction as the lava cools. The columns are generally perpendicular to the surface of cooling.

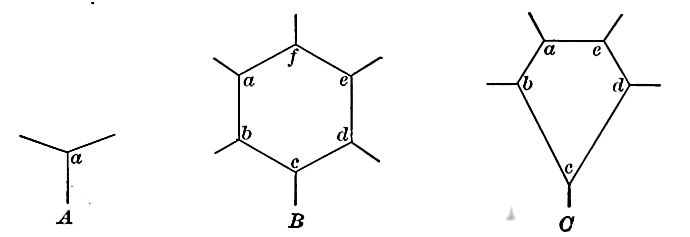
The explanation of the columns is probably somewhat as follows: The surface of the lava contracts about equally in all directions on cooling. The contractile force may be thought of as centering about equidistant points. About a given point, the least number of cracks which will relieve the tension in all directions is three (A,Fig. 8). If these radiate symmetrically from a point, the angle between any two is 120°, the angle of the hexagonal[p. 18] prism. Similar radiating cracks from other centers complete the columns (B,Fig. 8). A five-sided column would arise from the failure of the cracks to develop about some one of the points (C,Fig. 8). All igneous rocks are likely to be affected by cracks or joints, which run through them in various directions, but joints are not peculiar to igneous rocks.

[p. 19]
Pyroclastic rocks have somewhat the structure of sedimentary rocks. If the fragmental volcanic matter accumulates on the surface of the land, it. often lacks distinct stratification; but if it falls or is washed into water, it may be assorted and stratified. In this case it is distinguished from clastic rock by its constitution.
Minor structural and textural features of igneous rock. Most igneous rocks are made up of interlocking crystals of different sorts. These crystals may be so small that they are not readily distinguished by the eye, or they may be so large as to be easily seen, or some may be large and some small. If they are large enough to be distinct to the eye even without close scrutiny, the rock is coarsely crystalline. It is proposed to call all such rocks phanerites. In the case of phanerites, the interlocking of the crystals is evident (Fig. 9) . If the crystals are so small as not to be readily seen except by the help of a magnifying glass, the rock is an aphanite. In all igneous rocks, the crystals are of somewhat unequal size; but in some, there are certain crystals, usually of some one mineral, which are much larger than the others — so much larger as to be conspicuous. The rock is then said to be porphyritic (Fig. 10). The smaller crystals in which the larger ones are set may be so small as not to be readily distinguished (aphanitic), or they may be separately visible (phaneritic) .
[p. 20]
Some igneous rock looks like dark colored glass. Such rocks are indeed glass, and, like manufactured glass, were solidified from a liquid. Volcanic glass (obsidian) is one phase of solidified lava. It is formed when the liquid lava solidifies quickly, before the crystals have time to grow. Some igneous rock is made up partly of glass and partly of crystals, and between the rock which is all glass and that which is all crystals there are all gradations. Whether the lava becomes glassy or crystalline on hardening, or whether it is partly the one and partly the other, depends on the conditions under which it solidifies. All liquid lava contains the materials out of which crystals may be formed, under proper conditions.

Glassy and partly glassy rock may be compact or porous. Porous rock of the type shown in Fig. 11 is called scoriaceous. Rock of this sort is really lava froth, solidified, and the pores arc the spaces occupied by gases when the lava hardened. The bubbles were sometimes large and sometimes small. Pumice is porous volcanic glass, the pores being small.
Besides these varieties of texture which originate as lava hart lens, there are the textures peculiar to pyroclastic rocks. When quantities [p. 21] of volcanic dust, etc., (sometimes called volcanic ash) become coherent, as by cementation, the resulting rock is called tuff (or volcanic tufa). If the constituents are largely coarse, instead of fine, the resulting rock is volcanic agglomerate. Pyroclastic rocks are less abundant than other volcanic rocks.
The nature of lava. We commonly think of lava as melted rock, but this view is hardly the correct one. Lava is rather a solution of mineral matter in mineral matter. A simple analogy may illustrate what is meant. If pounded ice and salt are mixed together at a temperature of 30° F., the two form a liquid, though the temperature is too low to melt either one. We commonly say the salt is dissolved, but it would be just as correct to say that the ice is dissolved. The ice did not melt, because the temperature was too low to melt it. The two minerals, ice and salt, are dissolved in each other, and the solution takes place at a temperature below the melting point of either. Something of the same sort appears to take place when rock becomes liquid. The liquid form is probably assumed at a temperature below that which would be necessary to melt the minerals which make up the lava.
The propriety of regarding lava as a solution of minerals in minerals is shown in another way. Granite, an igneous rock, is made up chiefly of three minerals, quartz, feldspar, and mica. These minerals melt at .different temperatures. If lava were nothing more than mineral matter melted, the several minerals should solidify from the liquid as it cools in the reverse order of their fusibility. Of the above minerals, quartz would be the last to melt, and if lava were merely melted rock, quartz should be the first to take the solid form as the lava cools, and it should become solid when the lava reaches the fusing temperature of this mineral. But this is not what happens. The least fusible mineral may be the last to take the solid form. In other words, the order in which the various minerals solidify from the liquid lava is independent of their fusibility. This would not be the case if they were simply melted.
The liquid lava is essentially a fluid glass. It is analogous to common glass, which is a silicate of potash, soda, or other base, except that manufactured glass is relatively free from iron and other coloring substances which abound in the lavas, rendering them [p. 22] dark and more or less opaque. Lavas, too, are usually mixtures of several silicates, while manufactured glasses consist of only one, or at most a few. Furnace slag is essentially an artificial lava.
Solidification and crystallization. When a lava is cooled quickly, the commingled silicates solidify in their diffused condition essentially as they were in the liquid; for there is no time for the molecules of one kind to come together in regular systematic order, as is necessary to form crystals. The essential feature of crystallization is this systematic arrangement of the molecules of a given kind, according to a definite plan, giving a specific crystal form.
In a thick viscid liquid, this systematic arrangement of molecules into definite crystal forms takes place slowly, for the crystalline force in the silicates is far less energetic than that in water, which crystallizes into ice rapidly and with great force. Because of the slowness of crystallization, the solidification of the lava may catch the process of crystallization at any stage. If the lava is cooled quickly, the result is a glass; if less quickly, part glass and part crystals; if slowly enough, all becomes crystalline. In general, the slower the growth, the larger the crystals.
The crystal forms of the different minerals are unlike. Thus a crystal of quartz always takes the form of a six-sided pyramid (Fig. 51), if nothing interferes with its growth. The crystals of feldspar, mica, etc., take other forms (Figs. 49 and 50). All crystals forms are grouped into six (sometimes made seven) fundamental systems of crystallization, and there are a multitude of variations of special form in each. The treatment of these forms belongs to mineralogy.
The first crystals to form from a liquid lava tend to assume perfect forms, but when many crystals are forming at the same time, as is generally the case when lava solidifies, the crystals interfere with one another’s growth. The result is that they interlock in all sorts of ways, and the forms of most of the crystals are very imperfect.
Successive stages of crystallization. Since eruptions take pi .ice intermittently, it is obvious that cooling of the lava may be in progress beneath the surface during the intervals between eruptions. After a certain stage of partial crystallization has been reached [p. 23] during such time of quiet, a renewal of eruption may take place, and the whole mass of lava may be shifted into new surroundings, and a second phase of solidification may be superposed on the one already started. The rock may then show two phases of crystallization: (1) Large crystals of the kind or kinds which developed in the lava during the first stage of slow subterranean cooling; and (2) small crystals or glass developed during the more rapid cooling under the new conditions. The result is large crystals set in a matrix of small crystals or of glass. Rock of this physical composition is porphyry, a term which has a textural but not a mineralogical significance.
¶ Composition of Igneous Rocks
Nearly all the chemical elements known on the earth are found in igneous rocks, though but few of them are abundant. These few are regarded as the normal or essential constituents, while the rarer substances are regarded as incidental. The relative amounts of the more abundant elements in the crust of the earth, as nearly as now known, are shown in the following table:[1]
| Element | Symbol | Per cent in the Solid Crust |
|---|---|---|
| Oxygen | (O) | 47.02 |
| Silicon | (Si) | 28.06 |
| Aluminum | (Al) | 8.16 |
| Iron | (Fe) | 4.64 |
| Calcium | (Ca) | 3.50 |
| Magnesium | (Mg) | 2.62 |
| Sodium | (Na) | 2.63 |
| Potassium | (K) | 2.32 |
| Titanium | (Ti) | .41 |
| Hydrogen | (H) | .17 |
| Carbon | © | .12 |
| Phosphorus | § | .09 |
| Manganese | (Mn) | .07 |
| Sulphur | (S) | .07 |
It will be seen that only eight of the elements exceed one per cent, and no other one reaches one-half of one per cent. Many of the elements that are of the utmost importance in the affairs of men occur in quantities too small to be estimated in percentages. The precious metals, and even some of the more common ones, [p. 24] as lead, zinc, and copper, are of little importance quantitatively. We do not know just how the various elements of igneous rock are united in the liquid lava, but their combinations after the lava has become solid may be determined.
Union of elements. In a general study of the igneous rocks we may for the present neglect all but the first eight of the elements. Out of these elements come various chemical combinations when the lava solidifies; out of the combinations come the various minerals; and from the combinations of the minerals come various kinds of rocks. The union of oxygen with the other seven elements may be taken as a fundamental step in this series of combinations. The result is the following oxides: Silica (Si02), alumina (Al203), the ferrous, ferric, and magnetic oxides (FeO, Fe203, and Fe304), magnesia (MgO), calcium oxide (lime) (CaO), soda (Na20), and potash (K20). The oxygen sometimes unites in proportions different from those here given, but exceptions may be neglected here.
Of these nine oxides, silica acts as an acid, or more strictly as an acid anhydride. All the rest, except the magnetic oxide of iron, and sometimes the oxide of aluminum, act as basic oxides. The proportion of silica in igneous rocks is so significant that all such rocks are sometimes divided into three groups, as follows: Those with more than 65% of silica are acidic; those containing 55 to 65%, intermediate; and those containing less than 55% basic.
The union of silica (Si02) and lime (CaO) forms calcium silicate, CaO,Si02, or CaSi03. The union of silica and magnesia forms magnesium silicate, MgO,Si02, or MgSi03. Corresponding unions of silica and the other oxides named, give rise to other silicates.
Formation of minerals. Since but one of the leading oxides (silica) that abound in the average lava plays the part of an acid, a very simple conception of the general nature of igneous rocks may be reached by noting that they are mostly silicates of the seven leading basic oxides; that is, the oxides of alumina, potash, soda, lime, magnesia, and iron. This general idea is a very useful one, and represents a most important truth; but in its use we must not forget that there are many exceptions. Sulphur, phosphorus, chlorine, and other elements unite with the bases to form sulphates, sulphides, phosphates, phosphides, chlorides, etc. So also there [p. 25] are many minor bases that form silicates; and these minor bases unite with the minor acids to form many more or less rare minerals. Again, there are native metals in some igneous rocks. But altogether these minor compounds hardly reach more than one or two per cent of the whole.
There are, however, two exceptions of more importance. In the liquid lava the acid and basic elements are not always evenly matched. When there is an excess of silica, a portion remains free and takes the form of quartz (SiO2). If there is an excess of the basic oxides, the weakest one is usually left out of the combination. This is commonly the iron oxide, which then usually takes the form of magnetite (Fe3O4). It is a singular fact that quartz often forms when there is no excess of silica, and magnetite when there is no excess of base. Quartz (free acid anhydride) and magnetite (free basic oxide) sometimes occur in the same rock. The explanation of this is yet to be found. The oxides of silicon and iron form rather important exceptions to the general statement that igneous rocks are made up mostly of silicates, but, thus qualified, the statement expresses the essential truth.
Sources of complexity. But here simplicity ends, and the sources of complexity are several. In the first place, silica unites with the bases in different ratios, and thus gives rise to uni-silicates or ortho-silicates (ratio of oxygen of base to o"xygen of silica, 1:1), sub-silicates (the above ratio more than 1), bisilicates (ratio 1:2), tri-silicates or poly-silicates (ratio 1:3 or higher), etc. All the bases are not known to combine in all these ways, but many do in more than one.
If the silica united with each of the bases by itself alone, the results would still remain comparatively simple; but instead, it sometimes unites with two or more at the same time. Thus we may have an aluminum-calcium silicate. Not only this, but the different silicates may crystallize together in the same mineral. Thus a crystal may be made up of alternating layers of different silicates. As such alternations are not governed by any known mathematical law, there is no determinate limit to the number of combinations that may arise.
As a result of all this fertility of combination, the total number [p. 26] of silicious minerals in igneous rocks is large. Geology deals with these minerals as constituents of the earth, but only a few of them are so abundant as to require special notice here. It may be remarked also that, as they occur in the rocks, only a few of them can be identified by simple inspection, partly because some of them look much alike, and partly because the crystals are often minute and intricately intermixed.
Minerals of igneous rocks. Fortunately for the simplicity of geological study, a few minerals make up the great mass of the igneous rocks. These few are quartz, the feldspathic minerals, the ferro-magnesian minerals, and the iron oxides.
Quartz is the free acid already mentioned. Some of its leading characteristics are given on p. 80. Crystals of quartz are sLx-sided prisms, normally, capped by six-sided pyramids (Fig. 51); but in igneous rocks their forms are usually very imperfect.
The feldspathic group of minerals embraces those formed by silica in union with alumina, together with either potash, soda, or lime, or two or more of these together. The feldspathic minerals are normally light in color, ranging from white to red, gray, or greenish. The feldspars are minerals of the first importance in igneous rocks. The varieties of feldspar are given on p. 77.
The ferro-magnesian group embraces minerals formed by the union of silica with iron, magnesia, and lime, together with more or less of the other basic oxides. The ferro-magnesian minerals are normally dark (commonly greenish) from the presence of iron, the great coloring element of rocks; but the color distinctions do not hold good in detail, and cannot be safely trusted as a means of identification.
Among the ferro-magnesian minerals the most important are the pyroxenes (p. 80), the amphiboles (p. 75), and the micas (p. 79). The pyroxenes and amphiboles have nearly the same chemical composition, but differ in crystal form and physical properties. Hornblende (an amphibole) has been melted, and on cooling under proper conditions found to take on the form of augite (a pyroxene). Pyroxene is sometimes altered into uralite, one of the amphiboles. The pyroxenes and amphiboles are among the most abundant of the dark minerals in crystalline rocks.
[p. 27]
The two leading micas are the iron-magnesia mica, biotite, or black mica, and the potash mica, muscovite, or white mica, the familiar “isinglass” of the stove-door. Chemically, muscovite should go with the potash feldspars, but it is distinguished from them by its crystalline habit and physical properties. Chemically, too, biotite should go with the pyroxenes and amphiboles, which it closely resembles except in the form and properties of its crystals.
Two iron oxides, magnetite (Fe3O4) and hematite (Fe2O3), are widely disseminated in igneous rocks, but they are far less abundant than the silicates. They constitute the free bases already mentioned.
Summary of salient facts. The salient facts are, therefore, (1) that out of the 70-odd chemical elements in the earth, eight form the chief part of it; (2) that one of these elements uniting with the rest forms nine leading oxides; (3) that one of these oxides acts as an acid and the rest as bases; (4) that by their combination they form a series of silicates of which a few are easily chief; (5) that these silicates crystallize into a multitude of minerals of which again a few are chief; and (6) that these minerals are aggregated in various ways to form rocks. Possessed of these leading ideas, we are prepared to turn to the consideration of some of the conditions under which these combinations take place in the formation of rocks from liquid magmas.
¶ Classification of Igneous Rocks
Several features are involved in the current classification of igneous rocks Some of these features have been noted already, but they may be recapitulated here. All fragmental igneous rocks are pyroclastic, and pyroclastic rocks may be tuffs, agglomerates, etc. (p. 21). Rock formed by the solidification of lava without the development of crystals, is obsidian, if not porous. If porous (hardened rock-froth), they are pumice, scoriaceous glass, etc. If the rock is largely glass, but partly of small crystals, it is sometimes called pitchstone, because its freshly fractured surface looks like pitch or resin. When scoriaceous rock has its cavities filled by minerals deposited from solution, the rock becomes an amygdaloid. Porphyry has already been defined (p. 23). All these names are based on texture, rather than on mineralogical or chemical composition.
Most igneous rocks are wholly crystalline, and are classified on the basis [p. 28] of their composition. Their chemical composition determines their mineral composition, and the naming of the rocks is based on the minerals they contain. It will be seen, however, that the chemical composition is the more fundamental. The number of varieties of igneous rock is very large, but only a few of the more important need be mentioned here.

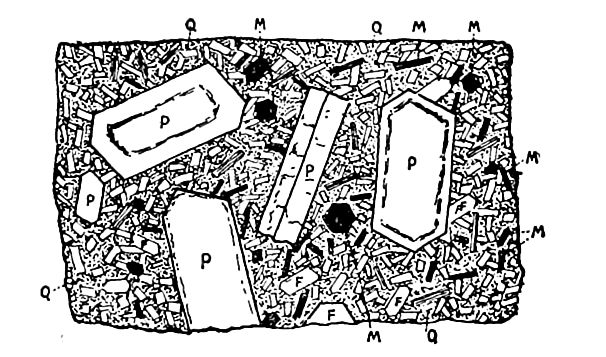
The granites. This name was originally used to designate a granular, i. e., a distinctly crystalline, rock, and it is still popularly and properly so used. In scientific treatises it has usually been confined to a rock composed chiefly of crystals of quartz, feldspar (especially orthoclase), (p. 79), and mica. It has recently been proposed to give it again a more general application, though not quite its original one, by including under it all phaneritei (p. 59) composed chiefly of quartz and feldspar of any kind, with mica, hornblende, or other minerals in subordinate amount. In normal granite, the crystals are distinct and sometimes large (Fig. 9). They are intimately mingled with one another, and in growing! interfered more or less with eaen other and so became interlocked. The granites are among the most common and easily recognized of the phanerites. Their color is determined largely [p. 29] by the feldspar, the red and pink varieties of the mineral giving rise to red granite, and the whitish varieties to gray granite.
Granites vary widely from their type form by the addition and substitution of other minerals, and these sometimes become as prominent as the type minerals. Thus other feldspars sometimes take the place of the orthoclase (p. 79), or accompany it; hornblende and other minerals take the place of the biotite, or occur with it, and so on. Whenever one of these replacing or accessory minerals is abundant, its name is often prefixed, as hornblendegranite. Granite grades almost insensibly into other types of igneous rock, as syenite, diorite, etc. Variations also arise from the absence of one of the three leading minerals.
The granites were formed from a lava rich in silica (68-70%), alumina, potash, and soda, but generally poor in lime, iron, and magnesia. Incidentally other substances are present. Granite is generally an intrusive massive rock which solidified slowly under conditions which permitted complete crystallization, with the development of large crystals.
When rock of the composition of granite is banded, it is gneiss (p. 84). The texture of graphic granite (see pegmatite, p. 86) is notably peculiar, due to the simultaneous crystallization of the quartz and feldspar (Fig. 12).
The syenites. The term syenite (from Syene on the Nile, where this sort of rock occurs) is now applied to a rock consisting essentially of feldspar and hornblende (with or without mica) ; but there is a complete gradation from the granites to the syenites. The syenites are richer in iron and magnesium than the granites, and poorer in silica (about 58-60%). The syenites also grade into other classes of rock as do the granites, and are named by similar prefixes, as augite-syenite, etc. The syenites are red or gray, according to the color of the feldspar, and are usually darker than the granites, which they resemble. The texture of syenite is like that of granite.
The diorites. The diorites are rocks which crystallized from lavas having about the same amount of silica as the lavas of the syenites, but poorer in the alkalies, and richer in the earthy bases. In current usage, diorite is defined as a rock composed of an intimate mixture of crystals of hornblende and a plagioclase feldspar. It differs from syenite in having plagioclase feldspar (p. 77) instead of orthoclase. By substitutions and the addition of accessory minerals, the diorites grade toward the granites and syenites on the one hand, and toward the gabbros on the other.
The gabbros. The name gabbro was formerly applied to a coarse-grained basic rock consisting of labradorite (p. 78) and diallage (p. 76); but the name has been extended gradually until it embraces a large group of rocks whose principal minerals are plagioclase (normally labradorite) and pyroxene (normally diallage), with magnetite or ilmenite (titanium iron oxide). The gabbros are usually dark colored and rather heavy. The pearly luster of the cleavage faces of the diallage gives a peculiar sheen to a fresh surface of the rock, in many cases.
[p. 30]
The Peridotites. These stand at the basic end of the series, having been formed from a magma in which the silica was low (39-45%), as were also the alumina, lime, and the alkalies, but in which the magnesia was high (35-48%). The rock consists very largely of the mineral olivine (a magnesium-iron silicate), associated with pyroxene, magnetite, and other very basic minerals. Little or no feldspar is present. The peridotites are much less abundant than the preceding classes.
Closely allied to the peridotites are rocks which are made up largely of a single basic mineral, as augite, pyroxenite, hornblendite, rocks essentially formed of the minerals augite, pyroxene, and hornblende, respectively.
The basalts. The term basalt is used in a somewhat comprehensive way for dark, compact, igneous rocks that appear to be nearly homogeneous owing to the smallness of the crystals which are usually so minute as to be identifiable only under the microscope. The leading minerals are plagioclase (usually labradorite or anorthite) and pyroxene (usually augite), with olivine and magnetite or ilmenite usually present. There is a considerable range in chemical nature, but the basalts are relatively poor in silica, and usually in potash and soda, but rich in lime, magnesia, and iron. The basalts are classed as basic, and are sometimes highly so. The lavas of basaltic flows were especially fluid, and spread out in thin sheets when poured out upon the surface. In cooling, basalt is prone to take on a columnar structure (p. 17). The columns of Giant’s Causeway and Fingal’s Cave are familiar examples.
The dolerites. The basalts graduate insensibly into the dolerites; indeed the dolerites may be regarded as basalts of coarser crystallization. The minerals are evident to the eye and range up to medium size. In the growth of the minerals, one crystal frequently encloses others. The dolerites have many varieties, due either to accessory minerals, or to the development of some of the constituents more amply than the others. The type may be said to consist of plagioclase and augite, the other minerals being regarded as accessories. The varieties are usually designated by prefixes, as olivinedolerite, etc., but special names are also used for some varieties.
The ancient dolerites have usually undergone internal changes and such rocks are often called diabases. While the use of the term has not been uniform, it accords with the better practice to regard the diabases simply as partially altered dolerites and basalts.
General names. The difficulty of distinguishing many of the foregoing rocks from each other by any means available in the field, owing to the minuteness of the crystals, and to the gradation of one type of rock into another, makes it desirable to employ certain general names which will correctly express the leading character of the rock without implying a knowledge of the precise mineral composition. A convenient term of this kind is greenstone, which merely indicates that the ferro-magnesian minerals are prominent, and usually give a greenish or greenish-black cast to the rock. The green [p. 31] stones embrace the diorites, dolerites, some of the gabbros, and the basalts, and may even extend to the peridotites and perhaps to others. Another convenient name is trap, which may be used for any dark, heavy igneous rock, such as basalt. The term basalt is sometimes used in much the same way.
Varieties of rock dependent upon conditions. From what has preceded, it is clear that the chemical nature of the liquid magma determines the mineralogical composition of the rock, if it is crystalline. But it may now be pointed out that the same lava which made a plutonic granite, might have made a porphyry, an obsidian, a pumice, or a tuff, under other conditions of cooling and hardening. The same is true of other varieties of the phanerites.
¶ The Disruption of Igneous Rocks
At the surface, igneous rocks are subject to mechanical disruption, and to chemical change which results in decay.
Mechanical disruption. The great agent of mechanical disruption is change of temperature. Heating by day and cooling by night produce some such change in rock as that effected by the sudden heating of cold glass or the sudden cooling of hot glass.

The heating of the surface of the rock by the sun expands it, and since the outer part is heated and expanded more than that below, a strain is set up between the superificial part and that below, and this strain may be enough to break off the outer part. “Shelling off” [p. 32] is frequently seen in the bowlders of the field (Fig. 13) and on the slopes of the hills and mountains (Fig. 14). Quick and great changes of temperature are more important than slow and slight ones in rock breaking. Thus annual changes are of little consequence, as compared with daily changes. If the changes of temperature involve a range above and below the freezing point of water, they may be still more effective, for if the pores and cracks of the rock are full of water, its expansion on freezing may break the rock. Great changes of daily temperature are found especially in high and dry regions, and it is in such places that rock breaking due to changes of temperature is most effective. Swift streams and waves, especially when they carry rock fragments, may also break up igneous rock which is so situated as to 1><> attacked by these agents of destruction, and the growth <>i* coots in cracks may contribute to the same end. All mechanical disruption of igneous rock leaves the fragments essentially like the original rock in composition.

Chemical disintegration. The silicate minerals which make up the larger part of all igneous rocks are usually complex, chemically. [p. 33] Not rarely they contain as many as three or four basic elements, in union with oxygen and silicon. It is well known that substances which are complex chemically, are, as a rule, less stable than those of simple constitution. Complex silicates, such as the feldspars, the micas, the amphiboles, and the pyroxenes (p. 80) tend to break up into simpler substances. The chemical changes are helped along by the oxygen, the carbon dioxide (C02) , and the water vapor of the air, as well as by water after it is precipitated. Some of the simpler changes may be noted.
Oxygen may enter into combination with the iron of a silicate mineral which contains iron. The iron is thus taken out of its silicate combination, and in union with the oxygen forms iron oxide, a simple and stable chemical compound. This process is oxidation. Oxidation affects other elements also.
Similarly the carbonic dioxide of the air may enter into combination with the base of a silicate mineral. Thus it enters into combination with the calcium of a mineral which contains calcium, taking the latter out of its combination with silica. The union of the calcium and the carbon dioxide gives rise to calcium carbonate. Magnesium and iron may be taken out in the same way, forming magnesium carbonate and iron carbonate, respectively. This process is called carbonation and the carbonates thus formed are simple and stable in composition. The carbonates are more soluble than most other common mineral substances.
Water may enter into combination with mineral matter, and the union is hydration. Thus when iron rusts (oxidizes), it is not merely oxygen which enters into combination with the iron, but water also. Iron rust is a hydrated oxide of iron.
The changes outlined above may be illustrated by the changes which take place in the decomposition of a complex mineral, such as augite, the composition of which is represented by the formula 2Ca0.2MgO.2FeO.Al2O3.Fe2O3.6SiO2. If to this be added 6CO2 and 2H2O, the products, after carbonation and hydration might be expressed thus: 2CaO. 2MgO. 2FeO.Al2O3. Fe2O3.6SiO2+6CO2 + 2H2O = 2CaCO3 + 2MgCO3 + 2H2O . Al2O3.2SiO2 + 2FeCO3 + Fe2O3 + S4iO2.
Oxidation, carbonation, and hydration, involving respectively the addition of oxygen, carbon dioxide, and water, increase the [p. 34] volume of the mineral matter. The result is that the rock affected crumbles. Thus the iron rust formed on a knife blade crumbles off. So the iron rust formed when oxygen and water unite with the iron in the rock, causes the rock in which the change takes place to crumble, partly because of the expansion involved.
Again, some of the simple compounds, especially the carbonates, formed when the rock decays, are somewhat soluble and may be dissolved and taken away. This tends to make the rock less compact by taking away one of its ingredients.
Oxidation, carbonation, and hydration therefore not only change the chemical nature of the rock, but they change its volume, allow some of its material to be carried off in solution, and in many cases cause it to fall to pieces. The .result is decayed rock — or one variety of rock waste. It is to be observed that the rock waste which arises from decay is unlike the original rock in composition. Some things have been added, and others taken away. In this respect, the waste sediment arising from decay is unlike that arising from rock breaking.
The products of decay may remain where they are formed, or they may be taken away. If they remain where formed for long periods of time, they may come to make a thick mantle of residual earth. The decayed rock is scores of feet in depth in many places, and hundreds of feet in some places. Chemical decomposition is greatest in warm regions, and the products of decay are least readily removed where there are forests. The products of decay are therefore likely to be deepest in warm, forested regions. They are very deep, for example, in some parts of Brazil.
¶ Sedimentation and Sedimentary Rocks
Removal of decayed rock. The breaking-up of igneous rock prepares the way for other processes, for the loose material which results from the disruption may be blown away by the wind, washed away by running water, or moved by any agency which shifts loose materials about on the surface of the earth. If the products of rock disintegration are coarse, they may become gravel after being rounded by streams or waves. If the material is finer, say [p. 35] of the size of small grains, it is sand; if still finer, it is mud when wet, and dust when dry.
Deposition of sediment. When carried by any transporting agency, such as wind or water, rock waste becomes sediment, and sooner or later is deposited as such. Some of the material picked up and transported by running water is left at the bases of the slopes of mountains and hills from which it is washed, and some of it is left on the flats through which streams flow; but much of it is carried to the sea and left there. The coarser part of the sediment carried to the sea is left near the shore, and the finer parts are taken farther out. This is seen along many coasts where the gravel of the shore-line grades out into sand, and this into mud as distance from the water’s edge increases. Thus it comes about that the coarser materials are more or less perfectly separated from the finer.
When the disintegration of the parent rock is by decay, the fine products are usually of different composition from the coarser. Thus the quartz grains of the granite are generally large enough to be readily seen individually; and as the rock decays, this mineral, already a simple compound, undergoes little change, and the grains remain in the rock waste. By moving water, they are rounded into the sand grains with which we are familiar. On the other hand, the crystals of feldspar, which has a complex composition, decompose into very fine particles (of kaolin or clay, Al2O3. 2SiO2-2H2O) unlike the feldspar in composition, and containing but a few of the elements of the feldspar. Thus it happens that the coarse materials, such as quartz, are chemically unlike the finer materials, such as the clay. Running water and waves assort the materials on the basis of their size; but the result is often the separation of materials which are chemically unlike. Thus beds of sand are accumulated, quite separate from beds of clay. They are separated in deposition because they are unlike physically, but in this case, physical unlikeness goes with chemical unlikeness.
Sediments which result from the mechanical breaking up of the rock are like the original rock in composition. When deposited as sand or gravel, such sediment might have about the same composition as the rock from which it was derived, if there was no [p. 36] decay. As a matter of fact, some decay usually accompanies mechanical breaking. The sediment which contains some of the feldspar of granitic rock is called arkose. Arkose represents incomplete decomposition of the parent rock.
Cementation of sediment into solid rock. After sediments such as gravel, sand, mud, etc., are deposited in the sea or elsewhere, they may be cemented into solid rock by the deposition of mineral matter held in solution in water. This cement binds the pebbles, the grains, and the smaller particles together, much as lime binds sand together in mortar. The cemented gravel makes conglomerate, or if the pieces of rock are angular, breccia; the cemented sand makes sandstone; and the cemented mud makes shale. These are common sorts of sedimentary rock. The cementation may take place while the sedimentation is in progress, or at a later time. Conglomerate, sandstone, and shale, made up chiefly of particles derived directly from other rock, are clastic rocks. Limestone may be broken up, and its particles redeposited and cemented again into solid rock. Such limestone is clastic, and limestone made of broken up shells, coral, etc., might also be regarded as clastic. In contrast with igneous rocks, clastic rocks are made up of particles of other rock, particles which were once separate and distinct, but now bound together by some sort of cement. The original constituents touch one another, but do not interlock, as do the crystals of igneous rock.
When sand, mud, etc., are deposited in the sea, shells of sea animals are frequently imbedded in them. If the shells or their forms are preserved, they make a record of the kinds of life that lived when the sediment was being deposited. If the sediments were deposited in lakes or on land, it is the shells or other relics of freshwater or land life which are found in them. All such relics of past life are fossils.
Non-clastic sediments. Not all sedimentary rocks are clastic. It has already been noted, in connection with the decay of rocks, that some of the compounds formed when rock decays are soluble. A considerable part of the materials dissolved are carried in solution to the sea. Here some of them are extracted by the animals and made into shells or other hard parts. When the animals die, their [p. 37] shells and other secretions are left behind. Shells and other secretions of calcium carbonate, cemented together, make limestone, the composition of which, when pure, is CaCO3. It is thought that most limestone was formed of organic secretions, though this is sometimes questioned. The shells, coral, etc., may or may not have been fragmented before cementation.
Limestone has many varieties. One variety is chalk (p. 82). Again, magnesium may replace the calcium in various proportions.

If there is any considerable amount of magnesium, the rock is called dolomite. The composition of ideal dolomite would be expressed by the formula CaMgCO3; but the amount of magnesium is variable. There was little magnesium in the shells, coral, etc., out of which much limestone was made. Sometimes the dolomization of the limestone (the conversion of CaCO3 into CaMgCO3) appears to have taken place long after the limestone was formed, while in other cases it appears to have taken place while the material of the limestone was being deposited.
Siliceous deposits. In the decomposition of igneous rocks, a little of the silica, as well as of the bases, is dissolved and carried away in solution. Certain organisms extract this from solution for their tests, shells, etc., just as others extract calcium carbonate. The accumulation of siliceous secretions often forms siliceous rocks.
[p. 38]
The diatom, radiolarian, and other oozes (Fig. 15) of the deep sea are the great examples. Sometimes layers of infusorial earth (tripolite) are made up of the secretions of diatoms and other aquatic organisms, accumulated in rather shallow waters. The most familiar examples of indurated rock formed in this way are certain flints and cherts that occur in limestone and chalk, chiefly as nodules, but sometimes in distinct beds.
Precipitation from solution. Sedimentary rock is formed in still other ways, as by direct precipitation from water which is saturated. Thus limestone and dolomite might be formed by direct precipitation from water if it became saturated with CaCO3 and MgCO3, and some limestone has been formed in this way. Rocksalt has been deposited in thick beds at various times and places, as it is being desposited now about Salt Lake in Utah. The sodium of the salt (composition NaCl) doubtless came from decaying rock, for many igneous rocks contain a little sodium in some complex combination. In the decay of the rock, the sodium is taken out of its complex combination, and made into some soluble compound, and then taken to the sea or to a lake. Its union with chlorine makes common salt. Gypsum (CaSO4) is another form of rock deposited in a similar way. Iron ore often occurs in such large bodies that it must be called rock, and some of these bodies of ore are formed by precipitation from solution. Salt, gypsum, limestone, and iron ore are peculiar among rocks, in that but one mineral enters into their composition when they are pure.
Coal is a sort of rock formed by the accumulation of vegetable matter. Some other sedimentary rocks, as noted above, are formed organically, though they can hardly be said to be organic.
The following table gives the principal classes of sedimentary rocks:
| Mechanically formed Clastic |
Conglomeratic rocks, — gravel, conglomerate, breccia, etc. Arenaceous rocks, — sand, sandstone, some arkose, etc. Argillaceous rocks, — clay, shale, etc., somelime stones. |
| Chemically formed Non-clastic |
Some carbonate rocks, e.g., travertine, siderite. Chloride rocks, — especially rock-salt. Sulphate rocks,— especially gypsum. Some siliceous rocks, — some cherts, etc. |
| Organically formed Non-clastic |
Calcareous rocks, — most limestones. Siliceous rocks, — siliceous oozes, sinter, etc. Carbonaceous, — coal, etc. |
[p. 39]
Original structure of sedimentary rocks. As originally deposited, the gravel, sand, mud, etc., are in beds. The beds may be of different material, or all may be essentially alike. In the latter case, there are often thin partings of some different material. Thus layers of sandstone may be separated by films of mud, layers of shale by a little sand, etc. These partings may record stages when the water was rougher or quieter than usual, or when it had an excess or a paucity of sediment.

At the time of their deposition, the beds of sediment conformed in a general way to the slope of the bottom where they were deposited. Since the slope of the sea bottom near shore is very gentle as a rule, the beds of sediment are, in most cases, nearly horizontal at the time of their deposition. Their angle of slope is rarely so much as 20°, and is commonly less than 5°.
Secondary structure of sedimentary rocks. Many sedimentary rocks have lost their original position through crustal movements, [p. 40] so that beds which were once horizontal now dip; that is, they depart from horizontally. The beds of sedimentary rock may even be on edge (Fig. 18), having a dip of 90°. The beds of a given region may all dip in one direction, or the dip may change from point to point. They may be folded, and the folds may be open (Fig. 16) or closed (Fig. 17) . These diverse positions in which strata are found are the result of disturbance subsequent to their deposition.

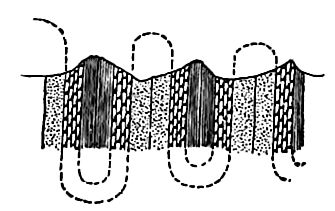
Besides being folded, the beds of sedimentary rock are often jointed (Fig. 2) the same as igneous rocks. Slipping may take place along the joint planes, producing faults (Fig. 19). The joints of sedimentary rock may be filled by the deposition of mineral matter from solution, making veins (Fig. 20). Other secondary [p. 41] structures of sedimentary rocks will be mentioned in connection with metamorphic changes.


[p. 42]
¶ Metamorphism and Metamorphic Rocks
We have already seen that igneous rocks undergo physical and chemical changes, whereby they are disintegrated, giving rise to what has been called rock waste; but the waste from one generation of rock is the raw material for rock of a new generation. It is “rock waste” in somewhat the same sense that lumber is forest waste.
Properly speaking, all changes which rocks undergo after being formed are metamorphic changes. According to this view, decayed rock is a phase of metamorphic rock; but it has been customary to limit the term “metamorphic” to those rocks which, instead of being disintegrated by the changes which they undergo, are rendered more compact, more complex in constitution, or more crystalline. Both sedimentary and igneous rocks may be metamorphosed.
Induration of sediments. The first step in the alteration of sediments is their induration, through the aid of cement, pressure, etc. Sandstone and shale are not commonly called metamorphic rocks, but they may be called metamorphosed sand, and metamorphosed mud, respectively. The cementing material of sediment is mineral matter deposited from solution in water. Thus material dissolved at and near the surface may be carried down by descending water, and deposited between the grains of sediment, binding them together. In the process of cementation, a striking plan is often followed, as illustrated by the cementation of sand by silica. It is to be remembered that the common grains of sand are more or less rounded particles of quartz, derived from the imperfect crystals of some rock which contained quartz. The crystallization of quartz, or any other mineral, involves the arrangement of the molecules in some definite way, peculiar to each mineral. When new silica is deposited from solution about these rounded grains of quartz sand, it is always deposited in such a way about each grain as to help to build it out into the form which crystals of quartz always take if free to grow as they will. Microscopic examination of well cemented sandstone often shows the original grains, distinct from the cementing material which appears as additions to original grains (Fig. 21). The enlargement of the [p. 43] grains may go so far that the spaces between the grains are filled. Sandstone thus changed becomes quartzite. Between loose sand at the one extreme, and quartzite at the other, there are all gradations. ’ Quartzite is often regarded as a metamorphic rock, but it is formed by a continuation of the process which cements sand into sandstone.
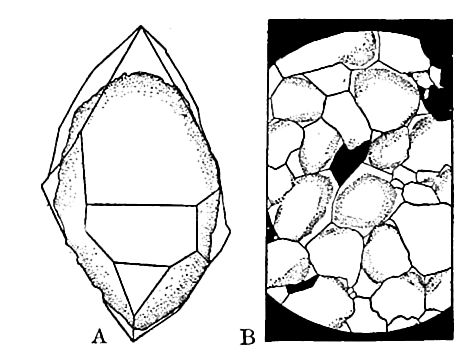
Grains of other minerals, also, such as feldspar, hornblende, etc., are subject to similar secondary enlargement. Important changes in rock are therefore brought about by solution, and then the re-deposition of dissolved mineral matter, through the influence of the water in the rocks. This process might be called aqueous metamorphism, because of the important part played by water. Since water is nearly always present in the rocks down to considerable depths, the changes which water produces are wide-spread, — are, indeed, nearly universal, down to the depths to which water penetrates, say five or six miles.
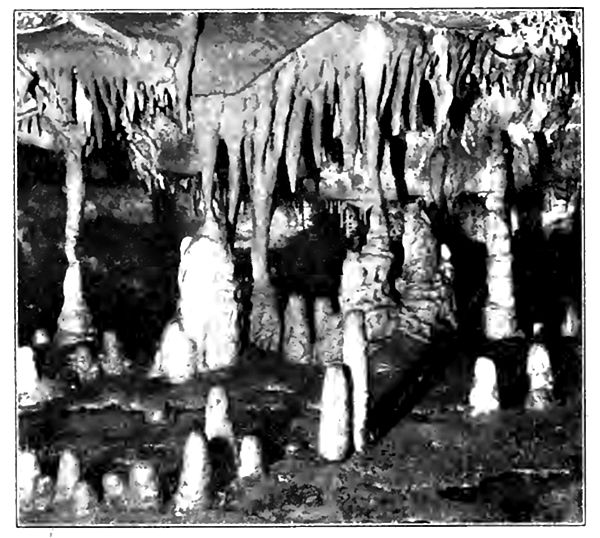
Cavity filling. Cavities in rock larger than pores also receive deposits, if the waters entering them carry mineral matter in solution. The making of veins (p. 40) is a case in point. The [p. 44] agates developed in some cavities afford another illustration. Here the successive layers are commonly silica (chalcedony, p. 76), and differ from each other in color and texture. Before the cavity is entirely filled, the deposit may change from chalcedony to crystals of quartz which grow with their pyramidal points toward the center of the cavity. Geodes are examples of a similar process in which the cavity is but partially filled with crystals (Fig. 22). The crystals of geodes are most commonly quartz or calcite, but they may be any other mineral that the waters are capable of depositing. Large cavities lined in tins way are known to miners as vugs, and these grade into caves with linings of crystals, and with stalactite and stalagmite (Fig. 23).
When small cavities1 in rock are filled by material deposited from solution, the result is sometimes called a secretion. Crystallined cavities (geodes,Fig. 22) and agates are examples of secretions. Crystal-lined cavities and veins are the same in principle.
Replacements. In both sedimentary and igneous rocks there are replacements, sometimes resulting in imitative or false forms. Thus the calcium carbonate of corals, shells, etc., may be replaced by silica. This substitution may take place in such a way that the minutest details of structure are preserved. Similarly, woody matter is sometimes replaced by silica, forming silicified or petrified wood.
Pseudomorphs. Again, the molecules of one crystal are sometimes replaced by different material, as the molecules of calcite by zinc carbonate, giving a pseudomorph of zinc carbonate after calcite. The zinc carbonate takes the form of the calcite, instead of the form which it would take if crystallizing under other circumstances. Hence the name. This sort of change may affect the crystals in any sort of rock.
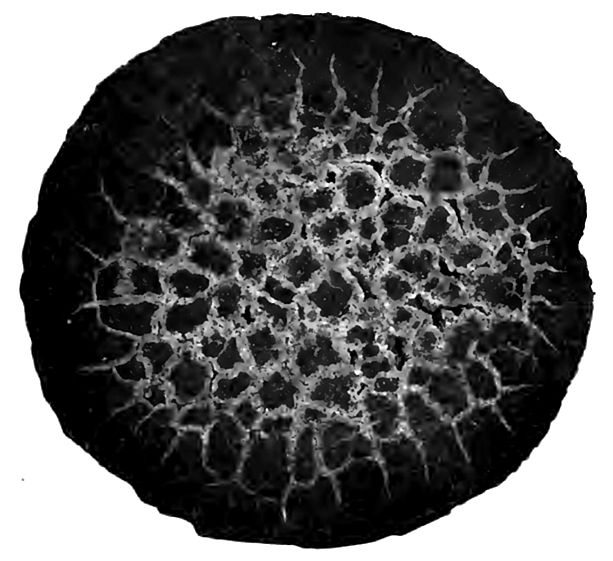
Concretions. A notable phase of the internal reconstruction of sedimentary rocks is the assembling of matter of the same kind. For instance, silica that was probably deposited in the form of siliceous shells and spicules of plants and animals, and disseminated through the sediments as they were deposited, is later aggregated into nodules of chert or flint (Fig. 24). Similarly, concretions of calcium carbonate or iron carbonate grow in silts [p. 45] or muds, pressing the clay back as they grow. In many other cases, too, kind comes to kind.

[p. 46]
In general, concretions are made by the deposition of mineral matter which was in solution, about a nucleus. The nucleus may be a leaf, a shell, or some bit of organic or inorganic matter. The material of the concretion may come from the immediately surrounding rock, or it may have been introduced from without, likewise by the agency of water. Concretions are generally of matter unlike that of the rock in which they form. Thus concretions of calcium carbonate are common in clay (Fig. 25), concretions of silica (chert) (Fig. 24) in limestone, and concretions of iron oxide in sandstone (Fig. 26).

Many concretions develop after the enclosing sediment was deposited. This is shown, in some cases, by the fact that planes of lamination may be traced through the concretions. Concretions also form in sedimentary rock during its deposition, and exceptionally, the rock is made up chiefly of them. The chemical precipitates from the concentrated waters of certain enclosed lakes sometimes take the form of minute spherules. From a fancied resemblance of these concretions to the roe of fish, the resulting rock was called oolite (Fig. 27). Oolite also forms in the open under proper conditions. It is now forming about some coral reefs, presumably from the precipitation of lime carbonate temporarily in solution. Some considerable beds of limestone are oolitic. The calcium carbonate of such rock may be subsequently replaced by silica, leaving the oolitic structure in siliceous rock. Beds of iron ore are likewise sometimes concretionary. Thus there are widespread beds of “flaxseed” iron ore made up of concretions of iron oxide which, individually, resemble the seed which has given the ore its name.
[p. 47]


[p. 48]
One of the most extraordinary features of some concretions of complex form is their symmetry. This may be of various phases; in exceptional cases there is a bilateral symmetry almost as perfect as in the higher types of animals.

Concretions sometimes develop cracks within themselves, and these may then be filled with mineral matter differing in composition or color from that of the original concretions (Fig. 28). Concretions the cracks of which have been filled by deposition from solution, are called septaria. They are especially abundant in some of the Cretaceous shales and clays. In not a few caaee the filling of the cracks appears to have wedged segments of the original concretion farther and farther apart, until the outer surface of the septarium is made up more largely of vein-matter than of the original concretion. The development of concretions in rock is not commonly looked upon as metamorphism, but it is really a metamorphic change in the broader sense of that term.
[p. 49]
In size, concretions may vary from microscopic dimensions to huge masses, 8, 10, or even more feet in diameter. The variations in shape are also great. The conditions of growth have much to do with the form. A concretion which starts as a sphere may find growth easier in one plane than another, when it becomes discoid. Two or more concretions sometimes grow together, giving rise to complicated forms.
Near the surface, the action of water commonly tends to the decomposition of the rock; but below a few hundred, or at most a few thousand feet, its general effect is to solidify the rock, for at these depths deposition exceeds solution, and oxidation, carbonation, etc., go on much more slowly than near the surface, or not at all. Thus oxidized and hydrated sediments may be buried to great depths, and under the pressure and perhaps the high temperature of these depths, deoxidation and dehydration may take place, with resulting diminution of volume.
[p. 50]
Incipient crystallization. A more pervasive metamorphic change in sedimentary rock is incipient crystallization. Some common limestones and dolomites are made up largely of small crystals, though the mass was originally a calcareous mud or ooze. New crystals are also developed in shales and other sediments, out of materials already present, or with such additions as groundwater may make. This process is a kind of incipient metamorphism, and takes place even under ordinary conditions of heat and pressure, through the agency of circulating ground-waters.
Change in chemical composition. Besides simple deposition in pores and cracks, the mineral matter in solution may enter into combination with other mineral matter, giving rise to new, and often to more complex and more compact mineral substances. The changes effected in this way go on slowly, but in the long course of time, they may go so far that none of the original rock material remains in its original condition — all having entered into new combinations. Soapstone or steatite, for example, is a rock composed essentially of such secondary material. Serpentine is a rock made up of a secondary mineral (serpentine) apparently derived from magnesian minerals. Chloritic rocks are rocks composed largely of the soft, green, hydrated mineral chlorite, derived from the pyroxene, amphibole, biotite, and perhaps other silicates of the parent rock. Other igneous rocks become talcose from the development of talc, a very soft, unctuous, hydrous magnesian silicate, developed from the magnesian minerals of igneous rock. All of these rocks may occur in large bodies. All are metamorphic rocks, developed primarily through the chemical rearrangement of the mineral matter of the original rock, with the addition of some matter brought in by ground water, and with the abstraction of some soluble materials from the original rock. The metamorphism may be said to be largely chemical.
By these and similar processes fragmental deposits are solidified into firm rock, and undergo internal changes which reorganize more or less the matter of which they are composed. The process is a very slow one usually, and takes place much more slowly under some conditions than under others. Some of the sands and muds of very early geologic ages are even now imperfectly solidified.
[p. 51]
Conditions favoring metamorphism. Besides water, two factors favor the metamorphism of rocks, viz., heat and pressure. Their action gives rise to three general cases, but these blend indefinitely: (1) great heat without exceptional pressure, (2) exceptional pressure without great heat, and (3) great heat and great pressure conjoined. Exceptional heat arises especially from the intrusion of hot lavas, and from pressure. Exceptional pressure arises chiefly from the weight of overlying rocks, and from lateral thrust due to the shrinkage of the globe. Thrust usually gives heat as well as pressure. The water in the rocks not only works changes in them, but greatly facilitates the chemical and mineralogical changes favored by heat and pressure.
Metamorphism by heat. When a mass of lava is poured out upon the surface, it bakes the mantle-rock which it overflows. The extent of the baking depends on the mass of lava and its temperature. The nature of the effect is much the same as in the baking of brick. It consists of dehydration of the material, induration of the loose matter by welding due to the partial fusion of the particles, and the development of new compounds. The time involved is short, the pressure trivial, and the water action limited. If the heat were sufficiently intense, the loose material over which lava flows would be fused; but complete fusion does not usually take place when lava spreads out on the surface.
Intrusions of lava (p. 15) heat the surface above themselves as well as that below. In this case the heat of the lava can only escape through the neighboring rock, and the temperature effects for a given mass of lava are more considerable. Furthermore, the time during which the adjacent rock is hot, and therefore the time during which thermal waters are operative is usually longer than in the case of extruded lavas, and the effects are chemical and crystalline changes, rather than mere baking. The resulting changes are greater the greater the mass of the lava and the higher its temperature. When a vent or fissure is the passageway for lavas that continue to come to the surface for a long time, as in the case of long-lived volcanoes, the rocks which form the walls of the vent are heated for long, and this gives rise to metamorphism through heat, without very unusual pressure, though usually with [p. 52] the free aid of water. In these cases the chief effects are chemical recombination and crystallization. In the limestones and sandstones the changes are simple, and in the shales more complex. In pure limestones and dolomites little chemical change takes place, but the molecules are rearranged into larger and more perfect crystals, making marble. The coarseness of the crystals is a rough sort of measure of the length of time during which the heat acts, and of its intensity, but much depends on the freedom of the attendant water circulation. Crystals an inch or two across are sometimes formed in the zone of contact between the intruded lava and the limestone where the attendant water action is important. If impurities, as silica, alumina, iron, etc., were present in the limestone, various silicate minerals (such as tremolite and actinolite, p. 74) may be formed in the marble. In pure quartzose sandstones, the effect is to cause the building up of the quartz grains until the interspaces are essentially filled, and the whole becomes a massive quartzite. Here, as in the marbles, impurities form adventious crystals, a very common one being hematite formed from the segregation of the ferric oxide of the sandstone.
In the shales, the material to be acted upon is more complex, for, while the main mass is composed of hydrous aluminum silicate, there is usually much free quartz, and often some potash, soda, iron, compounds of calcium, magnesium, etc., for the muds from which shales arise frequently contain not only the fully decomposed matter of the original crystalline rocks, but some fine matter worn from them by wind and water without decomposition. When this mixed matter is acted upon by high heat and moisture, it tends to return to its original crystalline state, so far as its changed composition permits. The result is the development of complex silicates, similar to those of igneous rocks, such as feldspar, mica, hornblende, etc. There is usually a predisposition to form mica in preference to other silicates if the proper constituents are present, and the result is that mica schists are common products of the metamorphism of shales by contact with bodies of lava. Mica schists are also formed in other ways, and other schists, dependent on the composition of the shales, are formed about intrusions [p. 53] of igneous rock. In all such cases pressure probably attends the heat, and is a factor in the development of the schists.
When the change induced by the heat is less considerable, the shale is baked, with incipient re-crystallization, and often takes the form of argillite, a compact, massive sort of shale.
Beds of hydrous iron oxide (limonite) or of iron carbonate (siderite) may be converted by heat into hematite or magnetite. Beds of peat, lignite, and bituminous coal are converted into anthracite by the driving off of the volatile hydrocarbons. If the process goes to the extreme, graphite is the result.


Metamorphism by pressure. Slaty structure. When rocks made up of clastic particles are compressed in one direction, and are relatively free to expand in others, the particles that are already elongated tend to turn so that their longer axes are at right angles to the direction of pressure, and all particles, whether elongate [p. 54] or not, are more or less flattened at right angles to the direction of stress. This may be readily seen where the particles are large (Fig. 29). As a result of the turning (or orientation) and flattening of their particles, rocks so affected split more readily between the elongate and flattened particles, than across them. In other words, the rocks cleave along planes normal to the direction of compression, and break with difficulty and with rough fracture across the planes of cleavage. The condition thus induced is known as slaty structure (Fig. 30), and is best illustrated by roofing-slate, which was originally a mud, later a shale, and finally assumed the slaty condition under strong compression. Sometimes the original bedding may still be seen running across the [p. 55] cleavage planes developed by pressure (Fig. 31). As the original mud beds were horizontal or nearly so, and as the thrust is most commonly horizontal or nearly so, the induced cleavage commonly crosses the bedding planes at a high angle. If the beds are tilted or bent before the development of the slaty cleavage, the angle between the original bedding and the slaty cleavage may be small.

Limestones, sandstones, and conglomerates are not so easily compressed as mudstones, and they usually take on only an imperfect cleavage normal to the direction of pressure.

Foliation, schistosity. More intense pressure in a given direction is capable of breaking down and deforming the most resistant rock. This must necessarily be attended with the evolution of [p. 56] heat, and thermal effects are mingled with pressure effects, but the thermal effects may be neglected for the moment. The first effect of the compression of such a rock as granite may be to crush it. It then becomes granular or fragmental, and is really a peculiar species of clastic rock (autoclastic) . By further compression, the fragmented material may be pressed into layers or leaves, much as in the development of slaty cleavage; but as a result of the nature of the material, the cleavage is less perfect. This is often attended by more or less shearing of the material upon itself. The result is a foliated or schistose structure (Fig. 4). A foliated structure may be developed thus in even the most massive rocks. Thus a granite may be transformed into a gneiss — which is like a granite in composition, but has a foliated structure; or a basalt may be converted into schist, a common term for foliated crystalline rocks. Porphyritic rock rendered schistose by pressure is shown in Fig. 32.
[p. 57]
Metamorphism by heat, pressure, and water. The mechanical results produced by pressure are always attended by the evolution of heat, and the heat and the pressure, in the presence of water, which is almost always in the rocks, greatly facilitate chemical changes. The result is that the mineral matter of the crushed and heated rock is often re-combined and re-crystallized. Under pressure, the new crystals arrange themselves so that their longest diameters are at right angles to the greatest pressure, and this orientation of the new crystals, like the orientation of other particles, helps to develop schistosity.
It is to be observed that two distinct processes may be involved in the making of schists. The one is the metamorphism of clastic rocks into crystalline schists, which may be regarded as an upbuilding process; the other is the mashing down of massive crystalline rocks into schists, which may be regarded as a descensional process. As a rule, neither process goes on alone in the development of schists. In both, there is more or less solution and rearrangement of the molecules, and in both there is probably something of crushing.
The kind of schist produced depends on the constitution of the rock metamorphosed. Thus basic rocks give rise to basic schists, and acidic rocks to acidic schists. It is obvious that ordinary shales cannot usually become basic schists, because in the production of the muds from which the shales are made, the bases were generally removed; but when shales are highly calcareous and magnesian, as when they grade toward limestone and dolomite, they may become basic schists (say hornblendic schists) by metamorphism. It is obvious that limestone and sandstone must retain largely their distinct composition.
Completion of the rock cycle. The crystallizing processes of metamorphism are fundamentally similar to the processes by which rocks crystallize from lavas; but in metamorphism, the work is done chiefly by the aid of an aqueous solution , while in the solidification of lavas the crystallization is from a mutual solution of the constituents in one another, where water was but an incident.
[p. 58]
¶ Various Classifications and Nomenclatures
From the foregoing sketch of the processes of rock-making it may be inferred that the varieties of rocks may be almost unlimited, and that they may be denned, named, and classified on many different bases; for example:
(1) If the mode of origin is chiefly in mind, rocks may be classed as igneous (lavas, tuffs, etc.); metamorphic (schists, gneisses, anthracite, etc.); aqueous (water-laid sediments, travertine, etc.); eolian (dunes, loess in part) ; glacial (till, moraines) ; clastic (mantlerock, sandstone, conglomerate, etc.); organic (peat, lignite, coal, etc., and indirectly, limestone, chalk, infusorial earth, etc.); and so on.
(2) If the textural characters are in mind, rocks are designated vesicular (pumice, scoria, etc.); glassy (obsidian); porphyritic (distinct crystals in obscure matrix) ; granitic (distinctly grained) ; compact, porous, earthy, arenaceous (sandy), schistose, etc.
(3) If the chemical composition is chiefly regarded, they may be classed as siliceous, calcareous, carbonaceous, ferruginous, etc.; or, if the chemical nature is considered, they are grouped as acidic, basic, or neutral.
(4) If the crystalline character is made the basis, they are designated phanerites (crystals distinct), aphanites (crystals very small), and amorphous (non-crystalline).
(5) If attention is fastened on certain ingredients, rocks are characterized as quartzose, micaceous, chloritic, talcose, pyritiferous, garnetiferous, etc.
(6) When rocks are regarded as mineral aggregates, the aggregates may be simple or complex. If simple, they are named from the dominant minerals, as dolomite, hornblendite, garnetite, anorthite, etc.; if complex, they take special names, as syenite, gabbro (p. 29), etc.
(7) When the point of view is structure of the mass, they arc classed as massive, stratified, shaly, laminated, slaty, foliated, etc.
(8) When physical state and genesis are considered, they are grouped as clastic, fragmental, or detrital (conglomeratic, brecciated, [p. 59] arenaceous, argillaceous, etc.); or pyroclastic (tufaceous, agglomeratic) , etc.
As sometimes one of these characteristics and sometimes another is most important in a given rock or in a given study, no one classification is satisfactory in all cases, yet each has its advantages in particular cases.
¶ A New System of Classification and Nomenclature
The familiar systems of classifying and naming rocks, if indeed they can be called systems, have grown up gradually out of earlier and cruder methods, many of which were inherited from popular usage. Most of the names and definitions came into use before modern methods of study were adopted. These systems, therefore, retain many crudities and inconsistencies, and lack adaptation to present needs and knowledge. They are too complex and difficult for field use and for general discussions, while not sufficiently exact and systematic for the more rigorous petrological discussions. A more adaptive and consistent system is needed, and in response to this need, a new system of classification of igneous rocks has been offered by a group of leading American penologists.[2] To some extent this proposed system may be extended to the metamorphic crystalline rocks, with necessary modifications and additions. The classification and nomenclature of the secondary rocks must probably always remain variable and plastic, to express the various points of view which it is desirable to take.
During the transition to this or some other new system, which seems inevitable, the appended alphabetical reference lists of the most common minerals and rocks, with brief definitions in accordance with current usage, will be found serviceable.
The proposed system includes two parts, a field system and a quantitative system, the one applicable to rocks on casual inspection, and the other, only after detailed study. The field system only is here outlined.[3]
The proposed field system. The proposed field names are based largely on texture and color. The mineral constituents are used for subdivisions when they can be determined; otherwise they are neglected.
Classifying chiefly on the basis of texture and crystalline state, there are three groups: Phanerites, in which all the leading mineral constituents can be seen without a lens; aphanites, in which all, or at least an appreciable part of the constituent minerals cannot be distinguished by the unaided eye; and glasses, in which the material is wholly or largely vitreous.
[p. 60]
I. The phanerites may be further classified by their chief mineral constituents as follows:
-
Granites (f. n.)[4], consisting largely of quartz and feldspar of any kind. with or without mica, hornblende, pyroxene, or-other minerals. This differs from the present common use in not regarding mica as an essential constituent, and in not distinguishing between alkali feldspars and calcic feldspars. The term therefore includes more than the term as heretofore used.
-
Syenites (f. n.), consisting predominantly of feldspar of any kind, with subordinate amounts of hornblende, mica, or pyroxene, but with little or no quartz. This differs from the common use in giving hornblende a subordinate place, and in embracing rocks with calcic feldspars.
-
Diorites (f. n.), consisting predominately of hornblende and subordinate of feldspar of any kind, with which there may be mica, pyroxene, or other minerals. This is nearly the present use, except that any kind of feldspar may be the subordinate mineral.
-
Gabbros (f.n.), consisting predominantly of pyroxene and subordinately of feldspar of any kind, with or without other minerals. This nearly coincides with one of the various present uses of the term except that the range of the feldspar is increased.
-
Dolerites[5] (f. n.), consisting predominantly of any ferromagnesian mineral not distinguishable as hornblende or pyroxene, with subordinate amounts of feldspar of any kind, and with or without other accessory minerals. A name to be used when the dominant mineral is clearly ferromagnesian, but cannot be satisfactorily identified by the eye, as either hornblende or pyroxene, although it may be probably one of these. In other words, the dolerites (deceptive) embrace the whole diorite-gabbro group when too obscure for separation.
-
Peridotites, consisting predominantly of olivine and ferromagnesian minerals without feldspar, or with very little.
-
Pyroxenites, consisting essentially of pyroxene without feldspar or olivine.
-
Hornblendites, consisting essentially of hornblende without feldspar or olivine.
II. The aphanites may be non-porphyritic or porphyritic.
(a) Non-porphyritic aphanites, when light-colored, may be classed as felsites; when dark-colored, as basalts.
(b) The porphyritic aphanites or porphyrias, when light-colored, arc leucophyrcs; when dark-colored, melaphyres. They may be classified further, according to the kind of phenocryst (distinct crystal) imbedded in the aphanitic ground-mass, as
Quartz-porphyries, or quartzophyres;
[p. 61]
Feldspar-porphyries, or felspaphyres (not felsophyres) ;
Hornblende-porphyries, or hornblendophyres; and so on.
These may be subclassed by color, as
Quartz-leucophyres, light-colored quartz-porphyries;
Quartz-melaphyres, dark-colored quartz-porphyries;
Feldspar-leucophyres ;
Feldspar-melaphyres; and so on.
III. The glasses are classified, according to color and luster, into obsidians or pitchstones when dark and lustrous; perlites when a spheroidal fracture gives them a pearly appearance; and pumice when greatly inflated by included gases.
In general discussions, it is regarded as serviceable to use the term granitoids in a broad generic sense, to include all crystalline rocks of the general granitoid type, including the granites, gneisses, etc. In a similar broad way, the term gabbroids may be used to include the dark crystalline rocks in which the ferromagnesian minerals predominate, as the diorites, gabbros, dolerites, peridotites, etc. In this convenient and comprehensive way, two contrasted groups of igneous rocks may be designated. As the granitoids are usually acidic and the gabbroids usually basic, the grouping represents a broad fact of importance.
¶ Structural Features Common to All Rocks
Certain structural features peculiar to one group of rocks or another have been mentioned. Thus stratification is characteristic of sedimentary rocks, columnar structure and massiveness of certain igneous rocks, foliation of many metamorphic rocks, etc. Joints also have been mentioned, and they affect all rocks, extending down to unknown, but often to great depths.
Faults. Slipping or displacement sometimes takes place along bedding, joint, or fracture planes, and such slippings are faults. The planes of slipping, the fault-planes, may be vertical, or inclined at any angle whatsoever. The angle of departure from the vertical is the hade. When the fault has a hade, the slipping may be such that the overhanging side goes down relatively (gravity or normal fault,Fig. 33), or such that it goes up relatively (thrust fault,Fig. 34) . The hade may approach or even reach 90°. Thrust faults are developed under lateral pressure. Thus faults developed during the folding of strata by lateral pressure are chiefly thrust faults, as in the Appalachian Mountains. Gravity faults imply [p. 62] crustal tension — not pressure. They were called normal, because they were supposed to be the most common. The fact seems to be that most faults of some regions are gravity faults, and most of those of some other regions are thrust faults; but both types may exist in the same region, though as a rule both types were not developed in the same region at the same time. The amount of vertical displacement is the throw of the fault. The amount of throw occasionally reaches several thousand feet.
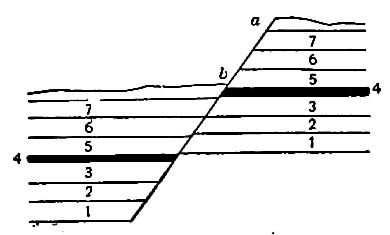


[p. 63]
If the slipping takes place between layers, instead of along joint planes, the term shear is commonly applied to the movement, but it is in reality a phase of faulting. Such faulting may take [p. 64] place when the beds are horizontal, or when they dip. When they are horizontal, the direction of the fault-movement is horizontal. The fault-movement may also be horizontal when the plane of slipping is vertical, or when it has any angle of hade. In this case, the fault shows itself at the surface by the displacement of straight lines, such as fences (Fig. 35). The fault at San Francisco in 1906 was of this type.
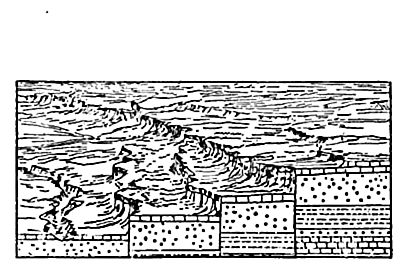
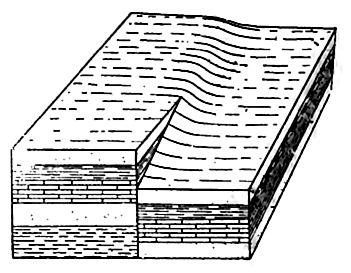
Where there is vertical movement in faulting, it is not usually possible to say whether the lower side has sunk, or the upper risen, or both, though in normal faults, sinking is probably the dominant movement. In any case, one side projects above the other, and the cliff is a fault-scarp. In the course of time the scarp is destroys I or obscured by erosion, and most faults are so old that this has taken place; but occasionally fault-scarps of mountainous heights are found, as along the east face of the Sierras, and along some of the basin ranges of Utah, Nevada, etc., but they are much modified by erosion (Fig. 36) . Great faults are probably the sum of numerous small slippings distributed through long intervals of time. Faulting is probably the most common cause of earthquakes.
Sometimes a fault branches, and sometimes the faulting is distributed among a series of parallel planes at short distances from one another, instead of being concentrated along a single plane, thus giving rise to a distributive fault (Fig. 37).
Faults are observed to die out gradually when traced horizontally, sometimes by passing into monoclinal folds (Fig. 38), and [p. 65] sometimes without connection with folding. Faults with great throw probably pass into folds below, in many cases at least (Fig. 39) .

The rock on either side of a faultplane is often smoothed as the result of the friction of movement. Such surfaces are slickensides. A slickensided surface has a slight resemblance to a glaciated surface, but generally gives evidence of greater rigidity between the moving surfaces. The rock along a fault-plane is sometimes crushed, making a crushed zone a few inches or feet wide. Such a zone becomes a passageway for ground-water, and so the site of ground-water work. Not a few such zones are “mineralized,” and have become the sites of important mines.
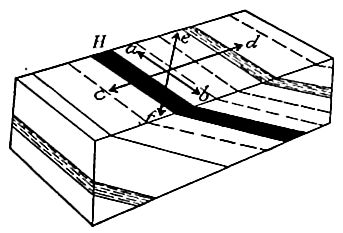
Effect of faulting on outcrops. Faulting may bring about numerous complications in the outcrops of rock formations. In a series of formations having a monoclinal structure (Fig. 40), many changes may be introduced. Let it be supposed in the following cases that, after faulting, the surface has been reduced to planeness by erosion. If the fault-plane is parallel to the strike of the beds (ab,Fig. 40), and hence a strike fault, the outcrop of a given layer (H) may be duplicated (Fig. 41), or it may be eliminated altogether (Fig. 42). If the faultplane is parallel to the direction of dip (cd,Fig. 40), a dip fault, the layer H will outcrop as in Fig. 43, if the downthrow was on the far side. The outcrops of H are offset, the amount of the offset decreasing with increasing angle of dip, and increasing with increasing throw of the fault. If the fault is oblique to the direction of dip and strike (ef,Fig. 40), an oblique fault, the outcrop of such a layer as H will have the relations shown in Fig. 44 if the down [p. 66] throw was to the left, and that shown in Fig. 45 if the downthrow was to the right. In the former case, it is said that there is offset with overlap; in the latter offset with gap. The amount of the overlap and gap, respectively, increases with the increase of throw and hade, and decreases with increase of dip. In all cases the outcrop (after the degradation of the upthrow side) is shifted down dip.
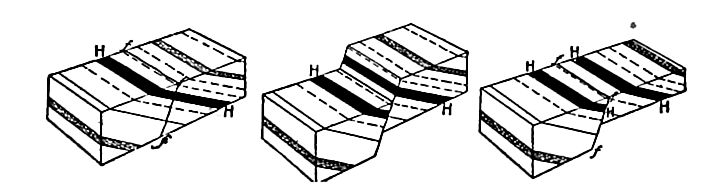
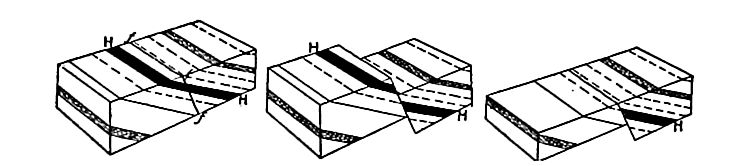
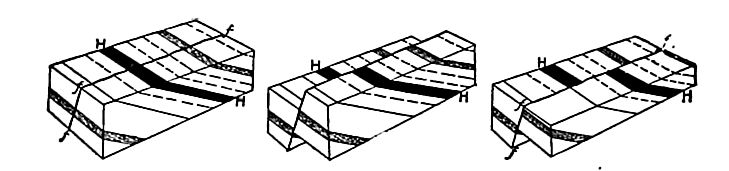
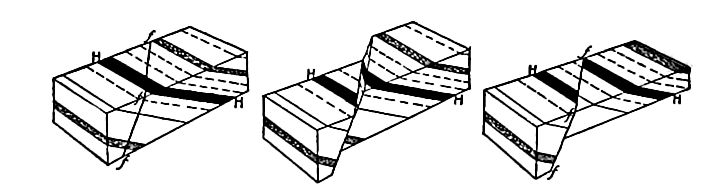
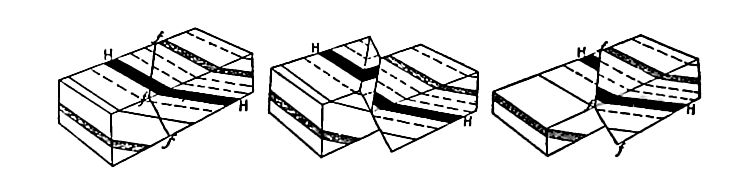
[p. 67]
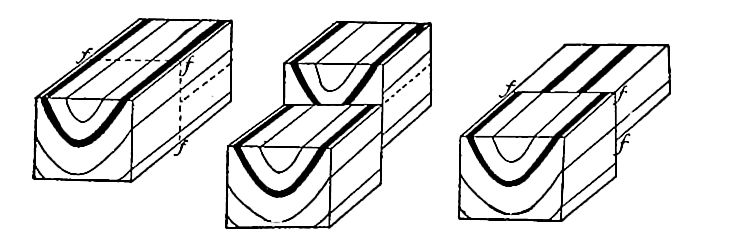
If a fault crosses folds at right angles to their axes, the effect is to change the distance between the outcrops of a given bed on opposite sides of the fault, after the truncation of the folded beds. The distance is decreased on the upthrow side of a syncline (Fig. 46) and increased on the upthrow side of an anticline (Fig. 47).
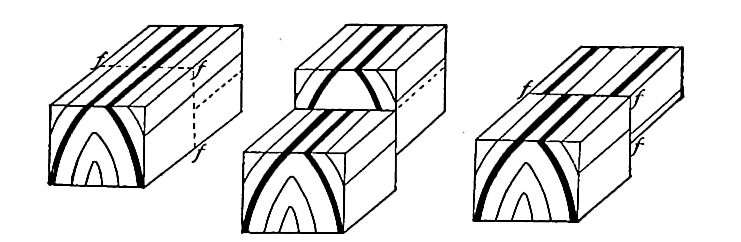
Various other complications arise under other circumstances. Since fault-scarps are not common, the detection and measurement of faults is usually based on the study of the relations of the beds involved, as illustrated by Figs. 41-47.
[p. 68]
¶ Ore-deposits
Ore-deposits are but a special result of the processes already discussed. They have peculiar interest because of their industrial value. An ore is a rock that contains a metal that can be profitably extracted, though for convenience the term is used more broadly to include unworkable lean bodies of ore material. The metal need not preponderate or form any fixed percentage of the whole, for the criterion is solely economic. A gold ore rarely contains more than a very small fraction of one per cent of the precious metal, while high-grade iron ore yields sixty-odd per cent of the metal. In iron ore, the metallic oxide or carbonate makes up nearly the whole rock; in gold ore, the metal is the merest incidental constituent from the petrologic point of view.
All the metals are disseminated through the rock substance of the earth and even throughout the hydrosphere, but they become ores only when concentrated in accessible places to a workable richness. Concentration of the metal is therefore the essential fact in the formation of ores. The degree of concentration required is measured by the value of the metal. The essential elements for consideration are, therefore, (1) the original distribution of the metallic materials through the rocks, (2) their solution by circulating waters (or, rarely, by other means), (3) their transportation in solution to the place of deposit, (4) their precipitation in concentrated form, and (5) perhaps their further concentration and purification by subsequent processes.
The few cases where ore-deposits are made by volcanic fumes or vapors may be neglected here, and only the more common phases of ore-deposition will be considered.
Original distribution of material. The original distribution of ore material through the primitive rocks is beyond the ken of present science, for even the nature of the true primitive rocks is unknown. For present purposes it is sufficient to regard all rocks concerned in ore-deposition as either igneous or sedimentary, and to inquire, first, how far ordinary igneous and sedimentary processes contribute to the segregation of ore material, and second, what the subsequent processes of local concentration are.
[p. 69]
Magmatic segregation. In a few instances workable masses of ore (e. g. iron) seem to have arisen by direct segregation in the liquid lava. It is not improbable that the segregation of metallic iron and nickel, and perhaps other metals may be a prevalent process in the deeper parts of the earth, giving rise to such masses of native iron as are found in basalt in Greenland. But masses of metal so segregated presumably gravitate downward, and have little relation to known ore-deposits. It is probable, however, that some segregation of metallic substances in the lavas that come to the surface, is a rather important first step. The rocks thus enriched perhaps determine the places where subsequent concentration is to take place. The acid igneous rocks (p. 24) are, on the whole, perhaps less rich in ore materials than the others; but there is no established law. It is probable that all sorts of lavas were richer in some places than in others at the time of their origin.
Marine segregation and dispersion. In the formation of the sedimentary rocks there was notable metallic enrichment in some cases, and depletion in others. The ground-waters of the land, after their subterranean circuits, carried to the seas various metallic substances in solution. In the main these substances appear to have been widely diffused, and to have been very sparsely deposited through the sediments, for sediments seem to contain less ore material than igneous rocks. But there are important exceptions to this general rule of sedimentary leanness.
The iron-ore beds of Clinton age ranging from New York to Alabama, and appearing also in Wisconsin and Nova Scotia, form a stratum in the midst of the ordinary sediments, and contain marine fossils. The great iron-ore beds of Lake Superior also were sedimentary in origin, and so were most other important iron deposits. It cannot be affirmed, in all cases, however, that the iron deposits were marine. In some cases, the ferruginous material was originally disseminated widely through land rocks, and was concentrated in the course of the sedimentary processes. In other cases it may have been derived from igneous rock rich in iron, extruded beneath water. In either case it is a sedimentary rock, of the non-clastic variety.
Limestone appears to have been enriched locally in lead and [p. 70] zinc, and more rarely in copper, in the course of its formation. The lead and zinc regions of the Mississippi basin have been regarded as regions of special subsequent enrichment, in areas where there was some enrichment at the time of sedimentation. This enrichment accompanying sedimentation has been attributed to solutions brought into the sea from neighboring lands, and precipitated by organic action in the sea-water.[6] This organic action may have been more effective in some areas than in others, because of the unequal distribution of life and the concentration of its decaying products. It is assumed that such precipitates were at. first too diffuse to be of value, and further concentration was required to bring them together into workable deposits.
Metallic material is sometimes concentrated to some extent in sand and mud in the processes of sedimentation, though more rarely. Copper-bearing shale is known, for example, in Germany, Texas, and elsewhere.
Since it is reasonable to suppose that land-waters, on reaching the margins of the water-basins, must occasionally find conditions favorable for the precipitation of their metallic contents, it is inferred that while the processes of sedimentation tended on the whole to leanness, they gave rise to (1) some very important oredeposits, notably the chief iron ores, the greatest of all ores in quantity and in industrial value, and (2) a diffuse enrichment of certain other areas which made them productive after. subsequent processes of concentration, while the sedimentary formations in general were left barren.
Origin of ore regions. From these considerations it appears that for the fundamental explanation of “mining regions” we must look mainly (1) to magmatic differentiation, so far as the country rock is igneous, and (2) to enrichment during sedimentation, so far as the rock is secondary. The subsequent processes in ore making consist in the further concentration of the ore material into sheets, lodes, veins, and similar aggregations by ground-water circulation, or else in the purification of the ores by the removal of useless or deleterious material.
[p. 71]
Surface concentration. The simplest of all modes of concentration takes place in the formation of mantle-rock. An insoluble or slightly soluble metallic substance sparsely distributed through a rock may be concentrated to working value by the decay and removal of the principal rock material, leaving the metallic matter in the residuary mantle. The tin ores of the Malay peninsula[7] are examples. Crystals of tin oxide were originally scattered sparsely through granite and limestone, but by the decay and partial removal of the rock, the crystals have accumulated in workable quantities. Certain gold fields and certain iron ores have acquired higher value in the same way; also certain ores of manganese, as those of Arkansas. Such residuary ores may be further concentrated by wash into gulches or alluvial flats, in the course of which the lighter parts of the mantle-rock are largely carried away, and the heavier, including the metal or its compounds, are mainly left behind. Gold placers are the best example. Such concentrates in past ages have in some cases been buried by later deposits, and hence certain ancient sandstones, conglomerates, and mantle-rocks have become ore-bearing horizons.
Purification. A somewhat different mode of concentration and purification has affected certain of the great iron deposits. As already explained, the iron compounds were originally parts of a sedimentary formation, and in beds. In some cases they were sufficiently pure, as first deposited, to be worked profitably; but in most cases they were seriously affected by undesirable mineral associates. After such impure deposits have been subjected to the percolation of waters from the surface for long periods, the impurities, under favorable conditions, are dissolved and taken away, and at the same time, new and valuable materials may be added. The great Bessemer ore-deposits of Lake Superior are examples. Originally impure carbonates or silicates, they have been converted into rich and phenomenally pure ferric oxides by ground-water. Vast quantities of lean ores lie in the tracts not thus purified and enriched. The waters seem to have brought in ferric oxide while they carried away the impurities, especially silica.[8]
[p. 72]
Solution and re-precipitation. Ore material is often leached out of the surface-rock by water circulating slowly through it, and carried on with the water until it reaches some substance which causes a reaction that precipitates the ore material. This substance may be a constituent of some rock which the circulating water encounters; but more commonly, the precipitation seems to be due to the mingling of waters charged with different mineral substances, the mingling inducing reactions resulting in the precipitation of the ore. Precipitation, however, does not necessarily follow such commingling. It is only when the mingling waters reduce the solubility of the ore material sufficiently, that deposition takes place. Changes of pressure and temperature also may enter into the process.
More concretely stated, the general process of underground ore formation appears to be this: The permeating waters dissolve the ore material disseminated through the rock, and carry it thence into the main channels of circulation, usually the fissures, porous parts, or cavernous spaces. If precipitating conditions are found there, deposition takes place. The precipitating conditions may be merely changes of physical state, such as cooling or relief of pressure, but probably much more generally they consist in the commingling and mutual reaction of waters that have pursued different courses, and aro differently mineralized. .
Location of greatest solvent action. Since solution precedes deposition, the location of the greatest solvent action may be noted. Water circulation is probably very slight below a depth of two or three miles at most, and above that depth there is little reason for supposing that the rocks of one horizon are more metalliferous than others of their kind. Thus there is no assignable reason why the igneous or sedimentary rocks at the surface are not as rich in ore material as the igneous rocks two or three miles below.
Solvent action is probably greatest where the temperature and pressure are highest, that is, in the deeper reaches of water circulation; but the amount of water passing in and out of the deeper zone is small compared with that of higher levels, and the total solvent action is quite certainly much greater in the upper zone than in the lower. At the same time, the solutions in the upper [p. 73] zone are quite certainly more dilute than those below. The horizon of greatest solution lies quite surely between the surface and a level slightly below the ground-water surface; or, in other words, in the zone where atmosphere and hydrosphere co-operate. Surface-waters are charged with atmospheric and organic acids and other solvents, and their general effect upon the rocks is markedly solvent down to or often below the permanent water-level. In this zone, concentration by residual accumulation may take place, as already noted, if the metallic compounds resist solution; otherwise this zone is depleted of its ore material by solution, and preparation is made’ for deposition elsewhere.
Solution also continues to take place varyingly as the water descends below this zone of dominant solution, and extends probably to the full depth of water circulation; but in the deeper circuit, precipitation also takes place, and with the waters taking up and throwing down material at the same time, it is difficult to estimate the balance of results. It is probable, however, that the result of these processes is to promote the development of the higher ore values at levels near enough the surface to be accessible, and along the main lines of ground-water circulation.
The influence of contacts. As ore-deposits depend on a dissolving state followed by a depositing state of the waters, and perhaps on a complex succession of these alterations, it is obvious that conditions which favor changes of state and the commingling of different kinds of water, are apt to be favorable to ore production. At any rate it is observed that many important ore-deposits occur at the contact between formations of different character. The contact of igneous rock with limestone is a rather notable instance. It is not to be inferred that such contacts are generally accompanied by workable ore-deposits, but merely that a notable proportion of workable ore-deposits occur at such junctions. It is rational to suppose that where the chemical nature of the two formations is in contrast, the waters that percolate through the one are likely to be mineralized very differently from those that course through the other, and hence that on mingling at the contact, reactions are specially liable to take place, and that when a valuable metallic substance is present, it is liable to be involved and, by chance, to [p. 74] suffer precipitation. Reactions are the more probable because the contact is likely to be a plane of crustal movement, and hence more or less open and accompanied by fractures, zones of crushed rock, and other conditions that facilitate circulation and offer suitable places for ore formation.
The effect of igneous intrusions. A special case of much importance arises when lavas are intruded into sediments that have previously been partially enriched in the ways above described. The igneous intrusion not only introduces new contact zones, and more or less fracturing, but it brings into play hot waters with their intensified solvent work, their more active circulation, and the reaction between waters of different temperatures. The special efficiency of these agencies is believed to be the determining factor in many cases. Furthermore the intruded lava may be rich in metallic substances, and so be a favorable site for later concentration, and the magmatic waters (waters from the lava) themselves appear to be a source of important ore-deposits. The present tendency is to attach more and more importance to the metallic contents of magmatic waters.
The influence of rock walls. The rock walls themselves are thought sometimes to be a factor in the reactions which precipitate ores. It appears that the effect of the wall may be to withdraw a constituent of the passing solution, and destroy its equilibrium in such a way as to cause the precipitation of the metallic constituent. Once deposited on the walls, ore aids the further accretion of ore of the same sort. The effect of the rock wall here noted is sometimes called mass action.
The special forms which ores assume in deposition, as beds, veins, lodes, stockworks, disseminations, segregations, etc., are chiefly incidental to the local situation in which the essential chemical or physical change takes place.
¶ Reference List of the More Common Rock-forming Minerals, and a Few of Economic Importance
Actinolite. A magnesium-calcium-iron amphibole (q. v.); commonly bright green to grayish green; crystals usually slender or fibrous.
Agate. A banded or variegated chalcedony quartz, (q. v.).
[p. 75]
Alabaster. A fine-grained variety of gypsum (q. v.), either white or delicately colored.
Albite. A soda feldspar (q. v.), an aluminum-sodium silicate; H.[9] 5-6; cleavage .perfect in two planes; luster vitreous or pearly white; occasionally bluish gray, reddish, greenish; sometimes opalescent.
Amethyst. A variety of quartz or corundum of purple or bluish-violet color, due probably to manganese.
Amphibole. The type of an important group of rockforming minerals known as the amphibole or hornblende group; a ferromagnesian silicate; monoclinic; H. 5-6; luster vitreous to pearly; fibrous varieties often silky; black, ranging through various shades of green to light colors ; embraces the magnesium-calcium varieties, tremolite and nephrite; the magnesium-calcium-iron variety actinolite; the aluminous-magnesium-iron-calcium variety hornblende, the most important member of the group, and others.
Andesine. A plagioclase feldspar (q. v.) ; a sodium-calciumaluminum silicate, intermediate in composition between albite and anorthite; H. 5-6; white, gray, grayish, yellowish, flesh red; luster sub vitreous, inclining to pearly.
Andalusite. An aluminum silicate; luster vitreous; whitish, rose red, flesh red, variety pearly gray, reddish brown, olive-green; H. 7.5, infusible; impurities sometimes so arranged in the interior as to exhibit a colored, crossed, or tesselated appearance in cross-section (chiastolite).
Anhydrite. A calcium sulphate; H. 3-3.5; luster pearly to vitreous; white, sometimes bluish or reddish; differs from gypsum in absence of water and in its greater hardness.
Anorthite. A plagioclase feldspar (q. v.); a calcium-aluminum silicate; varies much by impurities and admixtures; H. 6-6.5; pearly or vitreous luster; white, grayish, reddish.
Anthracite. Hard coal; hydrocarbon with impurities; supposed to be derived from bituminous coal by metamorphism.
Apatite. Essentially calcium phosphate with chlorine or fluorine; hexagonal; H. 5; luster vitreous or sub-resineous; colors usually greenish to bluish, characterized by a hexagonal form.
Aragonite. A calcium carbonate; differs from calcite in cleavage, and in being orthorhombic ; H. 3.5-4; luster vitreous or resinous; white, also gray, yellow, green, and violet.
Asphaltum. Asphalt; mineral pitch, bitumen; a natural mixture of different hydrocarbons; odor bituminous; melts at 90 to 100 degrees C. ; burns with a bright flame; graduates into mineral tars and through these into petroleum; probably the residue of the latter.
[p. 76]
Augite. One of the pyroxenes; an aluminum-calcium-magnesium-iron silicate; H. 5-6; monoclinic, crystals usually thick and stout; sometimes lamellar; also granular; black, greenish black, deep green; an important rock-forming mineral.
Beauxite. Essentially hydrated alumina; occurs in concretionary grains of clay-like form, whitish to brown; valuable as a source of aluminum.
Barite. Barites, heavy-spar, barium sulphate; orthorhombic, H. 5-3.5; luster vitreous to resinous, sometimes pearly; white, inclining to yellow, gray, blue, red, or brown; very heavy, sp. gr. 4.3-4.7.
Biotite. Black mica, a potash-aluminum-magnesium-iron silicate; monoclinic; easy basal cleavage into thin laminae; sometimes occurs as a massive aggregation of cleavable scales ; H. 2.5-3 ; luster splendent on cleavage surface; black to dark green; cleavage surfaces smooth and shining; a very common constituent of crystalline rocks.
Bitumen. The same as asphaltum (q. v.).
Calcite. Calcspar; calcium carbonate; rhombohedral, perfect rhombohedral cleavage; often taking the forms known as dogtooth spar, nail-head spar; frequently stalactitic and stalagmitic; H. 2.5-3.5; luster vitreous; white, occasionally pale shades of gray, red, green, blue, violet, yellow, brow n ; strong double refraction; embraces variety called Iceland spar; a very common mineral; the essential basis of limestone.
Chalcedony. A crypto-crystalline variety of quartz having a wax-like luster, either transparent or translucent; white, grayish, pale brown to dark brown, black, sometimes delicate blue, occasionally other shades; frequently occurs as the lining or filling of cavities, taking on a botryoidal or mamillary form.
Chlorite. The type of an important group of secondary minerals usually characterized by a green color, softness, and smoothness or unctuousness of feeling; they are usually aluminum-magnesium-iron silicates, with chemically combined water; derived from several other species, as pyroxene, amphibole, biotite, garnet, etc. ; embraces a number of species, among which are clinochlore, penninite, prochlorite, and delessite.
Chrysolite. Olivine; essentially a magnesium-iron silicate; orthorhombic; H. 6-7; luster vitreous; green, commonly olive-green, sometimes yellow, brownish, grayish green; highly infusible; a common constituent of certain basic igneous rocks; the name olivine is more commonly used by geologists.
Corundum. Alumina; an oxide of aluminum; H. 9; rhombohedral; large crystals usually rough; luster vitreous; color blue, red. yellow, gray, and nearly white; purer forms of fine colors are sapphires; the red variety is ruby, the yellow, oriental topaz, the green, emerald, and the purple, amethyst; dark colors, with iron oxide, emery.
Diallage. A variety of pyroxene (q.v.); H. 4; characterized by thin foliac; usually grayish green to grass-green, or deep green; luster on cleavage [p. 77] surface pearly, sometimes metalloid or brassy; an essential mineral in the gabbros, as sometimes denned.
Enstatite. One of the pyroxenes; essentially a magnesium silicate; orthorhombic ; H. 5.5; luster a little pearly on cleavage surface; metalloidal in the bronze variety (bronzite); grayish white, yellowish white, greenish white to olive-green and brown ; very infusible ; a common mineral in certain basic crystalline rocks.
Epidote. A complex aluminum-calcium-iron silicate of varying composition; monoclinic; H. 6-7; luster vitreous, pearly, or resinous; color usually pistachio-green, or yellowish green to brownish green; common in many crystalline rocks, usually as a secondary product.
The feldspars are aluminum silicates, with either potassium, sodium, or calcium, or two or more of these ; crystal systems, monoclinic and triclinic ; possesses distinct cleavage in two directions; H. 6-6.5; range in color from white through pale yellow, red, or green; and occasionally dark; triclinic feldspars frequently called plagioclase; the group embraces orthoclase, microcline, albite, oligoclase, andesine, labradorite, anorthite, and numerous varieties.
Fluorite. Fluorspar; calcium fluoride; isometric, usually cubic; H. 4; luster vitreous, sometimes splendent; white, yellow, green, rose, crimson red, violet, sky-blue, and brown; yellow, greenish, and violet most common; occurs usually in veins or cavities in beautiful crystalline form.
Galenite. Galena; lead sulphide; isometric, usually cubic; perfect cubic cleavage; luster metallic; lead-gray; a common ore of lead; occurs in veins and layers, also as linings of cavities.
Garnet. A complex silicate of varying composition, embracing aluminum, calcium, magnesium, chromium, iron, and manganese, but usually only two or three of these are present in abundance, and the varieties are characterized by the leading constituent; isometric, usually in dodecahedrons or trapezohedrons; H. 6.5-7.5; luster vitreous to resinous; commonly red or brown, sometimes yellow, white to blue, green or black; common in mica schist, gneiss, hornblende schist; also in granite, syenite, and metamorphosed limestone.
Glauconite. Green-sand, a hydrous potassium-iron silicate usually impure, amorphous, or earthy ; dull olive-green or blackish, yellowish, or grayish green; opaque, commonly occurs as grains or small aggregations.
Graphite. Plumbago, black lead; a form of carbon, usually impure; rhombohedral, but rarely appearing as a crystal; more often as thin laminae of greasy feel; yields a black adhesive powder; hence its common use for lead pencils; occurs in granite, gneiss, mica schist, crystalline limestone; sometimes results from alteration of coal by heat; occasionally occurs in basaltic rocks and meteorites.
[p. 78]
Gypsum. A hydrous calcium sulphate; monoclinic; perfect cleavage into smooth polished plates; occurs in a variety of forms, including fibrous and granular; H. 1.5-2; luster pearly and shiny; white, sometimes gray, flesh red, yellowish, and blue; impure varieties dark; crystallized varieties include selenite, satin-spar, alabaster, etc. ; easily recognized by its softness and want of effervescence with acids ; occurs in beds ; calcined and ground, constitutes plaster of Paris.
Hematite. Ferric oxide, Fe203; iron sesquioxide; rhombohedral, more commonly columnar, granular, botryoidal, or stalactitic; luster metallic, sometimes earthy; iron-black, dark steel-gray, red when earthy; gives red streak or powder; a leading iron ore, 70 per cent metallic iron when pure; the chief source of the red color of soils and rocks generally.
Hornblende. The most important amphibole (q. v.); name sometimes used as a synonym for amphibole.
Hypersthene. One of the pyroxenes (q. v.); a ferromagnesian silicate; orthorhombic ; H. 5-6; luster somewhat pearly on cleavage; surface often iridescent; dark brownish green, grayish, or greenish black and brown; a frequent constituent of crystalline rocks.
Iceland spar. A form of transparent calcite (q. v.)
Ilemnite. A titanium iron-oxide; rhombohedral; resembles hematite; luster submetallic; iron-black; powder black or brownish red; occurs frequently in crystalline rocks associated with magnetite.
Iron pyrites. Pyrite (q. v.).
Kaolin. Kaolinite; essentially a hydrous aluminum silicate; usually in clay-like or earthy form; white or grayish white; often tinged with impurities; commonly arises from decomposition of aluminous silicates, especially the feldspars; basis of pottery and china.
Labradorite. A plagioclase feldspar; essentially an aluminum-calciumsodium silicate; composition intermediate between that of albite and ;tnorthite; triclinic; H. 6; luster pearly or vitreous, gray, brown, or greenish; sometimes colorless or white; frequently shows play of colors; important constituent of various crystalline rocks, especially of the basic class; usually associated with a pyroxene or amphibole.
Leucite. Essentially an aluminum-potassium silicate, allied to the feldspars; H. 5-6; luster vitreous, white, ash-gray, or smoke-gray; occurs in certain volcanic rocks, particularly lavas of Vesuvius.
Limonite. Brown hematite, ocher; a hydrous iron oxide; commonly earthy; also concretionary, stalactitic, botryoidal, and mamillary, with fibrous structure; H. 5-5.5; luster silky, sometimes submetallic, but commonly dull and earthy; brown, ocherous yellow; streak and powder yellowish [p. 79] brown; constitutes ocher, bog-ore, ironstone, etc.; is the chief source of the yellow color of soils and rocks ; arises from the alteration of other iron ores.
Magnesite. Magnesium carbonate; rhombohedral ; white, yellowish, grayish white to brown; fibrous, earthy, or massive; found in altered magnesium rocks.
Magnetite. Magnetic iron ore; iron oxide, Fe304; octahedral or dodecahedral; strongly magnetic ; H. 5.5-6.5; abounds in igneous and metamorphic rocks.
Marcasite. White iron pyrites; iron sulphide; same composition as pyrite, which it closely resembles; H. 6-6.5; luster metallic, pale gray, bronze, or yellow; prone to decomposition; disseminated through various rocks, particularly plastic clays containing organic matter.
Mica. The type of an important group of rock-forming minerals well known for their perfect cleavage into thin elastic laminae ; among the leading varieties are the common potassium mica (muscovite), the sodium mica (paragonite) , the lithium mica (lepidolite), the magnesium-iron mica (biotite), the magnesium mica (phlogopite), and the iron-potash mica (lepidomelane).
Microcline. A triclinic feldspar, closely resembling orthoclase in appearance and having the same composition.
Muscovite. Common or potash mica ; essentially an aluminum-potassium silicate; H. 2-2.5; monoclinic; remarkable for its basal cleavage; splits easily into exceedingly thin, flexible, elastic laminae; luster vitreous, more or less pearly or silky; colorless or variously tinged brown, green, or violet; a common mineral in crystalline rocks, particularly in the granites or gneisses.
Oligoclase. A plagioclase feldspar; essentially an aluminum-calcium sodium silicate which may be regarded as a mixture of albite and a small amount of anorthite; triclinic; luster vitreous, pearly, or waxy; whitish grading into greenish and reddish; H. 6-7; common in crystalline rocks.
Orthoclase. A potash feldspar; essentially a potassium-aluminum silicate; varying by the replacement of the potassium by sodium and less frequently by other substitutions ; monoclinic ; occurring in distinct crystals and also in cryptocrystalline forms; cleavage planes perfect with pearly luster on cleavage surface; white, gray, and flesh-red, occasionally varying to greenish white and bright green; H. 6-6.5; difficultly fusible; sanidine a glassy variety; a very common mineral, especially in the granites, syenites, and gneisses.
Olivine. Chrysolite (q. v.).
Petroleum. Naphtha; a native mineral oil; a hydrocarbon, commonly believed to arise from organic matter, both animal and vegetable, but held by some to be due to deep-seated chemical and thermal action.
Plagioclase. A general term embracing the triclinic feldspars whose two cleavages are oblique to each other; embracing albite, oligoclase, andesine, labradorite, and anorthite (q. v.).
Plumbago. Graphite (q. v.).
[p. 80]
Pyrite. Iron pyrites, fool’s gold, iron sulphide; isometric; commonly in cubes; H. 6-6.5; luster metallic, splendent, or glistening; pale brass-yellow ; occurs widely disseminated throughout a large class of rocks ; usually harder and lighter in color than copper pyrites, and deeper in color than marcasite, which has the same composition.
Pyroxene. The type of a large and important group of rock-forming ferromagnesian minerals; varies in composition and embraces a large number of varieties; usually a magnesium-iron-calcium silicate; crystals usually thick and stout, but varying greatly; sometimes lamellar and fibrous; H. 5-6; luster vitreous inclining to resinous; green of various shades verging occasionally towards light colors, more often to browns and blacks; among the minerals belonging to the pyroxene group are augite, bronzite, diallage, diopside, enstatite, hypersthene, and others.
Quartz. Crystallized silica; hexagonal; crystals commonly six-sided prisms capped by six-sided pyramids; without cleavage; H. 7; hard enough to scratch glass; usually transparent, glassy, colorless when pure; shaded by impurities to yellow, red, brown, green, blue, and black; varieties, amethyst, purple or violet; false topaz, yellow; rose-quartz, smoky quartz, milky quartz, etc.; chalcedony is a cryptocrystalline (crystals not visible) variety; carnelian, a red chalcedony; agate, a variegated or banded chalcedony; moss-agate, a chalcedony containing moss-like or dendritic crystallizations of iron or manganese oxide; onyx, chalcedony in layers; jasper, an opaque, colored quartz, usually red or brown; flint, an opaque impure chalcedony; chert, an ill-defined term applied to an impure flinty rock; hornstone, a translucent, brittle, flinty rock.
Serpentine. A hydrous magnesium silicate; usually in pseudomorphie forms; also fibrous, granular, cryptocrystalline, and amorphous; H. 2.5-4; luster subresinous to greasy, pearly or earthy, resinous or wax-like; feel, smooth and somewhat greasy; leek-green to blackish green and siskin green verging into brownish and other colors; apparently derived most commonly [p. 81] from chrysolite or olivine, but also from other magnesian minerals; sometimes constitutes the bulk of rock masses.
Siderite. Iron carbonate; rhombohedral; H. 3.5-4.5; luster vitreous, more or less pearly, ash-gray, yellowish or greenish, also brownish; occurs as extensive iron deposits and in crystalline rocks.
Steatite. Soapstone, a variety of talc (q. v.) ; hydrous magnesium silicate.
Sulphur. A well-known element occurring native in volcanic regions; also formed by the decomposition of sulphides, particularly pyrites.
Talc. A hydrous magnesium silicate; usually in foliae; granular or fibrous forms; also compact; easy cleavage into thin flexible laminae, but not elastic; luster pearly on cleavage surface; apple-green to silvery white; H. 1-2; a secondary product from the alteration of magnesian minerals; distinguished by its soft, soapy feel, soapstone being one variety; whitish form is known as French chalk.
Topaz. An aluminum silicate, with part of the oxygen replaced by fluorine; orthorhombic ; H. 8; luster vitreous; colorless, straw-yellow verging to various pale shades, grayish, greenish, bluish, and reddish; distinguished by its hardness and inf usibility ; occurs in crystalline rock.
Tremolite. A calcium-magnesium amphibole; a common constituent of certain crystalline rocks.
Zeolites. A group of minerals derived from the alteration of various aluminous silicates.
¶ Reference List of the Commoner Rocks
Adobe. A fine silty or clayey deposit formed by gentle wash from slopes and subsequent lodgment on flats; especially applied to clayey accumulations in the basins and on the plains of the western dry region.
Agglomerate. An aggregate of irregular, angular or subangular blocks of varying sizes, usually of volcanic origin; distinguished from conglomerate, in which the constituents are rounded.
Alluvium. Sediment deposited by streams.
Amygdaloid. A vesicular igneous rock whose cavities have been filled with minerals deposited from aqueous solutions; the fillings are called amygdules, because sometimes almond-like in form.
Andesite. An igneous rock consisting essentially of the plagioclase feldspar andesine (sometimes oligoclase) and pyroxene (or some related ferromagnesian mineral) ; sometimes cellular, porphyritic, or even glassy ; usually rich in feldspar microlites.
Anorthosite. A rock consisting mainly of the feldspar labradorite.
[p. 82]
Aphanite. A rock whose constituents are so minute as to be indistinguishable to the naked eye; rather a condition of various rocks than the name of any specific rock.
Arenaceous rocks. Either those which are mainly sand, or those in which sand is a notable accessory.
Argillite. A clayey rock; usually applied to hard varieties only.
Arkose. A sand or sandstone formed of disaggregated granite or similar rock in which a notable part of the grains are feldspar or other silicate; sand when undefined, is understood to be quartzose.
Augitite. A rock made up mainly of augite.
Basalt. A dark, compact basic igneous rock consisting of a mass of minute crystals sometimes with more or less glassy base, often containing also visible crystals; composed of plagioclase and pyroxene, with olivine magnetite, or titaniferous iron as common accessories; a basic lava in which the crystallization has taken place rapidly; usually rich in crystallites or microlites; graduates into dolerite aud basic andesite.
Bituminous coal. Common soft coal, intermediate between lignite and anthracite; contains much bituminous matter, i. e., hydrocarbons.
Bowlders. Rounded masses of rock, such as those that have been shaped by glaciers.
Breccia. A rock composed of angular fragments, contrasted with pudding-stone or conglomerate, in which the fragments are rounded.
Buhrstone. A compact, flint-like silicious rock full of small cavities, so named from use as millstones.
Calc-sinter (calcareous tufa) . A loose cellular deposit of calcium carbonate made by springs; travertine is the better term, as tufa should be left for volcanic elastics.
Cannel coal. A very fine-grained homogeneous bituminous coal, giving off much gas and burning with a candle-like flame.
Chalk. A fine-grained soft rock composed essentially of calcium carbonate derived from minute marine organisms.
Chlorite schist. A schistose rock in which chlorite is a predominant mineral; usually greenish, whence the name.
Clastic rock. Formed from the debris of broken-down rocks; the same as fragmental or detrital rock.
Clay. A term commonly applied to any soft, unctuous, adhesive deposit, but in strict use confined to material composed of aluminum silicate; ma inso-called clays are chiefly silicious silts or loams.
Clay ironstone. A clayey rock heavily charged with iron oxide, usually limonite; commonly in concretionary form.
Clinkstone. A name applied to phonolite because of its metallic clinking sound when struck; composed of orthoclase, with nephelite and one or more of the ferromagnesian minerals as accessories.
[p. 83]
Chert. An impure flint, usually of light color, occurring abundantly in concretionary form as nodules in certain limestones.
Coal. A carbonaceous deposit formed from the remains of plants by partial decomposition.
Concretions. Aggregates of rounded outlines formed about a nucleus; the material is various: clay, iron ore, calcite, silica, etc.
Conglomerate (puddingstone). A rock formed from rounded pebbles; consolidated gravel.
Coquina. A rock formed almost wholly of small and broken shells; especially applied to a shell limestone of Florida.
Dacite (quartz-andesite). An andesite (q. v.) with quartz.
Diabase. A dolerite (q. v.) which has undergone alteration; consists essentially of plagioclase feldspar and augite, with magnetite or titaniferous iron as a common accessory; one of the greenstones.
Diatom ooze. A soft silicious deposit found on the bottom of the deep sea, made largely or partly of the secretions of diatoms.
Diorite. An igneous rock usually of dark-greenish color, consisting of plagioclase feldspar and hornblende; often speckled from the commingling of light feldspar and dark hornblende.
Dolerite. A fine-grained igneous rock composed of plagioclase feldspar (labradorite or anorthite) and augite (or related ferromagnesian mineral, as enstatite, olivine, or biotite), with magnetic or titaniferous iron as common accessories; crystals usually of medium size, assuming the ophitic structure; embraces many of the greenstones; graduates into basalt on the one hand and gabbro on the other.
Dolomite. A magnesian limestone. X Drift. In common American usage, a mixture of clay, sand, gravel, and bowlders formed by glacial agencies.
Eolian rocks. Deposits formed by wind, embracing especially dunes and much of the loess.
Felsite (felstone). A light-colored aphanitic rock composed of feldspar, often with quartz, in which the crystallization is very imperfect or obscure, giving a close-grained texture with conchoidal fracture and flinty aspect; certain varieties are called petrosilex and halleflinta.
Fire clay. A clay capable of standing a high degree of heat. The percentage of iron, calcium, magnesium, and the alkalies must be very low. Often found in association with coal.
Flint. A compact dark chalcedonic or lithoid form of quartz.
Freestone. A sandstone of uniform grain without special tendency to split in any direction.
Fulgurites. Glassy tubes, produced through fusion by lightning in penetrating sand, earth, or rock.
Gabbro (euphotide). A crystalline rock composed of the plagioclase [p. 84] feldspar labradorite (or anorthite), and diallage (or a related ferromagncsian mineral), with magnetite or titaniferous iron as a common accessory.
Gangue. A term applied to the crystalline material in which ores are imbedded.
Ganister. Essentially a quartz silt, pulverized quartz, or silicious fire clay suitable for lining iron furnaces.
Garnetite. A rock composed largely of garnets.
Geyserite. The silicious sinter deposited about hot springs.
Gneiss. A foliated granite, consisting normally of quartz, feldspar, and mica; the feldspar typically orthoclase.
Granite. A granular crystalline aggregate of quartz, feldspar, and mica; the feldspar typically orthoclase; popularly and properly used for any distinctly granular crystalline rock.
Granulite. A fine-grained granite with little or no mica.
Graphic granite. (See pegmatite.)
Greensand. A sand or sandstone containing a notable percentage of grains of glauconite.
Greenstone. A comprehensive term used to designate igneous and metamorphic crystalline rocks of greenish hue and of intricate and often minute crystallization; they are mostly dolerites, diabases, and diorites; a convenient term for field use where the constituents cannot be determined, and for general use when the variety is unimportant.
Greywacke. A sand rock in which the grains are largely basic silicates instead of quartz.
Hornblendite. A rock essentially composed of hornblende.
Hornstone. A very compact, silicious rock of horn-like texture, allied to flint; term also applied to flinty forms of felsite.
Hydraulic limestone. (See waterlime.)
Hypogene rocks. Those formed deep within the earth under the influence of heat and pressure.
Ironstone. A rock composed largely of iron, usually applied to clayey rocks having a large iron content.
Infusorial earth (tripolite). An earthy or silt deposit consisting chiefly of the silicious secretions of diatoms.
Itacolumite. A flexible sandstone whose pliability is due to an open arrangement of sand grains which are held together by scales of mien.
Jasper. A reddish variety of chalcedonic quartz.
Lapilli. Small fragments of lava ejected from volcanoes; volcanic cinders.
Lava. A molten rock, especially applied to flows upon the surface, whether from vents or from fissures; also applied to the solidified product.
Lignite, (brown coal). A soft, brown, impure coal.
Limestone. A rock composed primarily of calcium carbonate, though magnesium sometimes replaces a part of the calcium. (See dolomite and marble.)
[p. 85]
Loess. A very fine porous silicious silt containing some calcareous material which often collects in nodules (Loss Kindchen) or in vertical tubules; characterized by a peculiar competency to stand in vertical walls; held by some to be eolian, by others to be fluvial or lacustrine, and by still others to be partly eolian and partly aqueous.
Mantle rock. (See regolith.)
Marble. Typically a granular crystalline limestone or dolomite produced by metamorphic action ; but the term is variously applied to calcareous and even to other rocks that are colored ornamentally and susceptible of polish.
Marl. An earth formed largely of calcium carbonate, usually derived from the disintegration of shells; or the calcareous secretions of plants, notably the stoneworts ; term also sometimes applied to glauconitic and other fertilizing earths.
Melaphyre. A term of varying usage; most commonly applied perhaps to an altered basalt (q. v.), especially an olivine-bearing variety.
Meta-diabase. A term sometimes used for a metamorphic diabase; in like manner meta is prefixed to dolerite, syenite, etc.; not in general use.
Meta-igneous rock. A metamorphosed igneous rock.
Metamorphic rock. A rock which has been altered, particularly one which has been rendered crystalline, or recrystallized by heat and pressure in conjunction with water.
Meta-sedimentary rock. A metamorphosed sedimentary rock.
Microgranite. A very fine-grained granite.
Microlites. Incipient crystals found in glassy lavas; usually needleshaped, or rod-like; occurring singly and in aggregates.
Millstone. (See buhrstone.)
Minette (mica-syenite). A rock consisting essentially of orthoclase and mica, or a syenite in which mica replaces hornblende or predominates over it.
Monzonite. A granitic rock composed of orthoclase and plagioclase in nearly equal proportions, with ferromagnesian minerals ; a rock intermediate between syenite and diorite.
Mudstone. Solidified mud or silt, shale.
Nodules. Concretionary aggregations of rounded form.
Novaculite (hornstone, oilstone). A very fine-grained, hard sandstone or siltstone, used for whetstones.
Obsidian. A typical form of volcanic glass usually of the acidic class.
Onyx. A variety of chalcedonic quartz having colored bands alternating with white; the “Mexican onyx” is a crystalline calcium carbonate, variegated with delicate colors due to iron and manganese.
Oolite. A limestone or dolomite composed of small concretions resembling the roe of fish.
Ooze. A soft deposit of the deep sea; characterized usually by a microscopic shells from which it is mainly derived; as diatom ooze, globigerina ooze, etc.
[p. 86]
Peat. The dark brown or black residuum arising from the partial decomposition of mosses and vegetable tissue in marshes and wet places.
Pegmatite. A term of ill-defined usage applied to rocks whose grain varies from coarser to finer, and often takes on peculiar aspects due to the simultaneous crystallization and mutual inter-growths of the crystals; graphic granite is a distinct type of pegmatite in which quartz and orthoclase crystals grew together along parallel axes so that cross-sections give figures resembling certain Semitic letters (Fig. 12).
Peridotite. A very basic igneous rock composed chiefly of olivine with augite or related ferromagnesian minerals, with magnetite and chromite as accessories.
Perlite (pearlstone). A form of glassy lava made up in part of small spheroids formed of concentric layers which have a lustrous appearance like pearls.
Phonolite (nephelite-trachyte, clinkstone). A compact resonant igneous rock formed of sanidine and nephelite, with accessories.
Phyllite (argillite). A variety of indurated, partly metamorphosed, clay silt in which finely disseminated micaceous scales are abundant and lustrous ; intermediate between typical clay slate and mica-schist.
Pitchstone. A dark vitreous, acid, igneous rock of less perfect glassy texture than obsidian, and more resinous and pitch-like.
Plutonic rocks. Igneous rocks formed deep within the earth under the influence of high heat and pressure; hypogene rocks; distinguished from eruptive rocks formed at the surface.
Porphyry. A rock consisting of distinct crystals embedded in an aphanitic ground-mass, or of large crystals embedded in smaller ones.
Pumice. A glassy form of lava rendered very vesicular through inflation by steam.
Pyroclastic rocks. Fragmental or clastic rocks produced through igneous agencies, embracing volcanic ashes, tuffs, agglomerates, etc.
Pyroxenite. An igneous rock consisting essentially of pyroxene.
Quartzite. A rock consisting essentially of quartz, usually formed from quartzose sandstone by cementation or metamorphic action.
Regolith. A name recently suggested to embrace the earthy mantle that covers indurated rocks, chiefly residuary earths; mantle-rock.
Rhyolite. An aphanitic or partly glassy igneous rock showing flowage lines, usually applied only to the acidic varieties.
Sandstone. Indurated sand usually composed of grains of quartz, but not necessarily so; sometimes contains grains of the various silicates.
Schist. A crystalline rock having a foliated or parallel structure, split ting easily into slabs or flakes, less uniform than those of slate; they are composed mainly of the silicate minerals.
Scoriae. Light, cellular fragments of volcanic rock, coarser than pumice.
Septaria. Concretions the interior cracks of which have been filled with calcite or other mineral deposited from solution (Fig. 28).
[p. 87]
Serpentine. A rock consisting largely of serpentine; derived in most cases by alteration from magnesian silicate rocks.
Shale. A more or less laminated rock, consisting of indurated muds, silts, or clays.
Slate. An argillaceous rock which is finely laminated and fissile, either due to very uniform sedimentation or (more properly) to compression at right angles to the cleavage planes; e.g., common roofing-slate (Fig. 30).
Soapstone (steatite). A soft unctuous rock, composed mainly of talc.
Stalactites. Pendant icicle-like forms of calcium carbonate deposited from dripping water.
Stalagmites. The complement of stalactites formed by calcareous waters dripping upon the floors of caverns.
Steatite. (See soapstone.)
Syenite. A granitoid rock composed of orthoclase and hornblende, or other ferromagnesian mineral; the name was formerly applied to a granitoid aggregate of quartz, feldspar, and hornblende.
Till (bowlder clay). A stony or bowldery clay, or rock rubbish formed by glaciers.
Trachyte. A name formerly applied to a rock possessing a peculiar roughness due to its cellular structure; but at present mainly confined to a compact, usually porphyritic igneous rock, consisting mainly of sanidine associated with varying amounts of triclinic feldspar, augite, hornblende, and biotite.
Trap. A general term for igneous rocks of the darker basaltic types.
Travertine. A limestone deposited from calcareous waters, chiefly springs ; usually soft and cellular, and hence also called calcareous tufa; calc sinter.
Tuff (tufa). A term including certain porous granular or cellular rocks of diverse origins; the volcanic tuffs embrace the finer kinds of pyroclastic detritus, as ashes, cinders, etc. ; the calcarous tufas embrace the granular and cellular deposits of springs ; the better usage limits the term to volcanic elastics.
Waterlime. An impure argillaceous limestone possessing hydraulic properties.
Laboratory work. Laboratory study of the common rock-making minerals and of the common types of rocks should be undertaken in connection with this chapter. The groups of minerals mentioned on page 26 should be made familiar first. The identification of these minerals in the rocks should follow. The arrangement of the minerals is important in some cases, especially in metamorphic rocks. It is as important to stop these studies at the right point, in this connection, as to take them up. The technicalities of mineralogy, and the refinements of rock classification should not be touched , at this point. Appropriate limitations for the study of igneous rocks, at this point, are suggested on pages 27-30, and 59-62.
Clarke, F. W. Bull. 168, U. S. Geol. Surv. ↩︎
Cross, Iddings, Pirsson, and Washington. Quantitative Classification of Igneous Rocks. ↩︎
For a statement of the quantitative system reference should be made to the original work mentioned in the preceding foot-note. An abbreviated statement appears in the larger work of the present authors, Vol. I, p. 454, et seq. ↩︎
The initials f. n. (field names) are introduced to show that the term is used in the broad field sense proposed. ↩︎
Added by the authors of this work. ↩︎
Chamberlin. Geol. of Wis., Vol. IV, p. 599, et seq., 1882. ↩︎
Penrose. Jour, of Geol., Vol. XI, pp. 135-155, 1903. ↩︎
Van Hise & Leith, various monographs of the U. S. Geol. Surv. ↩︎
H. = Hardness. ↩︎
The following definitions are given, as nearly as practicable, in accordance with present common usage, which is, however, more or less varying and inconsistent. ↩︎