Public domain
[p. 195]
Many familiar facts demonstrate the presence of abundant water beneath the surface. The thousands of wells in lands peopled by civilized man, and the many springs which issue from the slopes of mountains and valleys are sufficient proof both of the wide distribution of ground-water and of its great abundance.
Certain well-known facts make it clear that ground-water is intimately connected with rainfall. The level of the water in wells commonly sinks during droughts, and rises after rains; and the sinking is greater when the drought is long, and the rise is more notable when the rainfall is heavy. Many springs flow with reduced volume in times of drought, and others cease to flow altogether. Furthermore, rain-water is seen to sink beneath the surface, wherever the soil is porous. Sinking through the soil to the solid rock, it finds cracks and pores, and through them it descends to greater depths. Nowhere are the rocks beneath the mantle rock so compact and so free from cracks, when any considerable area is considered, as to prevent the percolation of water through them. The conditions which influence the amount of water which sinks beneath the surface have been mentioned (p> 110).
Supply of ground-water not altogether dependent on local rainfall. The amount of ground-water in a given region does not always depend entirely on the local rainfall. Ground-water is in constant movement, and entering the soil or rock at one point it may, after a subterranean journey, reach a point far from that where it entered. Thus beneath the Great Plains of the West there is much subterranean water which fell on the eastern slopes of the Rocky Mountains. It has flowed beneath the surface to the plains where some of it is now drawn out for purposes of irrigation in [p. 196] regions where rainfall is deficient. The accompanying diagram (Fig. 164) illustrates the flow here described.
The ground-water surface. Water table. The water table has already been defined (p. 117) as the upper surface of the groundwater. In a flat region of uniform structure, the ground-water surface is essentially level, though it rises and sinks with the rainfall.
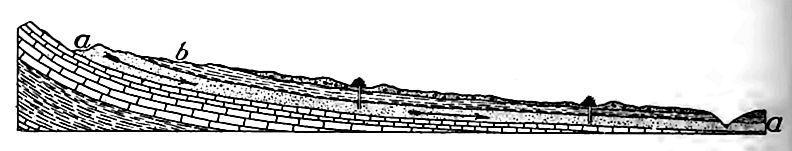
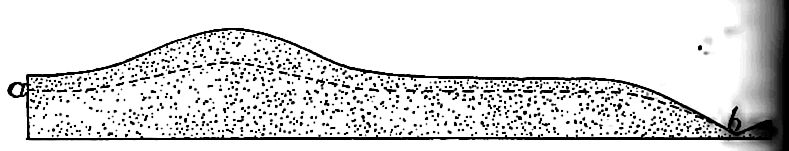
Where the topography of a region is not flat, the ground-water surface is not level. As a rule it is higher (though farther belo^ the surface) under an elevation than under surrounding lowlands, as illustrated by Fig. 165. The reason is understood readily, a hill of sand is exposed to rainfall, most of the water falling on ii porous surface sinks into it. If the precipitation continues lon$ enough, as in a protracted rain, the hill of sand will be filled with water, the water occupying the interstices between the grail The water in the hill tends to spread, but since the movement involves friction, the spreading is slow. With the spreading, tin surface of the water in the sand sinks, and sinks fastest at tin center where it is highest (Fig. 165). If no more water were added, the surface of the water in the hill would, in time, sink nearly the level of the water in the surrounding land; but at every stflj preceding the last, the surface of the water would be higher beneath the summit of the hill than elsewhere, though farther from the surface. In regions of even moderate precipitation the watersurface beneath the hills rarely sinks to the level of that in the lowlands about them, before it is raised by further rains.
[p. 197]
The water-surface beneath the lowlands also sinks. Some of it finds its way into valleys, some of it sinks to greater depths, and some of it evaporates; but since the water-surface beneath the elevations sinks more rapidly than that beneath the lowlands, the two approach a common level. Their difference will be least at the end of a drought, and greatest just after heavy rains.
Depth to which ground-water sinks. The depth to which ground-water penetrates has not been determined by actual observation. The deepest borings or excavations of any sort are little more than a mile deep, and at this depth there is nothing to indicate that the limit of water is being approached. There is a popular belief that water sinks until it reaches a temperature sufficient to convert it into steam; but except for special localities where hot lava lies near the surface, this belief does not appear to be well founded.
Assuming the temperature of water sinking beneath the surface to be 50° F., its temperature must be raised 162° to bring it to the temperature at which it would boil at sea-level. With this initial temperature, the following table shows the depths at which water would reach a temperature of 212° F. under various assumptions as to the rate of increase of temperature. It shows also the pressure in atmospheres which would exist at these several depths if the overlying rock were full of water.
| Rate of Increase of Temperature Initial temperature 50° F. |
Depth at which Temperature of 212° would be reached |
Equivalent Pressure in Atmospheres |
|---|---|---|
| 1° for 50 feet | 8,100 feet | 238 (approximately) |
| 1° for 75 " | 12,150 " | 357 |
| 1° for 100 " | 16,200 " | 478 " |
The temperature at which water boils increases with the pressure. A pressure of about 200 atmospheres is the critical pressure for water; that is, the pressure which, if increased, will prevent boiling altogether. The depth at which a pressure of 200 atmospheres [p. 198] would be reached, supposing the upper rock to be full of water, is about 6,800 feet. The temperature of the water at this depth, under various assumptions, is shown in the following table:
| Initial Temperature | Rate of Increase of Temperature |
Temperature at a depth of 6,800 feet |
|---|---|---|
| 50° | 1° for 50 feet | 186° Fahr. |
| 50° | 1° for 75 " | 141° " |
| 50° | 1° for 100 " | 118° " |
None of these temperatures is so high as the boiling-point of water at sea-level. It is therefore clear that at this depth, water has not even closely approached the boiling temperature for this depth, and since this is the depth of the critical pressure, it cannot boil at any greater depth, where the pressure would be greater. The descent of water is therefore not stopped, under normal conditions of crustal temperature, because it reaches its boiling-point. Locally, as in the vicinity. of active or recently extinct volcanoes, the case may be different.
It is conceivable that water may descend until it reaches its critical temperature, that is, the temperature which, if increased, will cause the water to become water-gas in spite of pressure. The critical temperature is somewhere between 610° and 635°. The depth at which the critical temperature would be reached, under various assumptions, is shown in the following table:
| Initial Temperature | Rate of Increase of Temperature |
Depth of Critical Temperature |
|---|---|---|
| 50° | 1° for 50 feet | 28,000 to 29,250 feet |
| 50° | 1° for 75 " | 42,000 to 43,125 " |
| 50° | 1° for 100 " | 56,000 to 57,500 " |
There is good reason, in the increasing density beneath the surface, and on other grounds as well, for believing that the rate of increase of temperature decreases with depth, and therefore that the rate of 1° for 50 feet for the depths concerned is far too high. The greater depths of the table above are therefore believed to more nearly represent the truth than the lesser ones. If this correct, the depth at which the critical temperature would be reached is probably not less than 50,000 feet.
[p. 199]
If descending water reached its critical temperature by going down to these great depths, the extent to which the resulting water-gas might be absorbed into the interior of the earth is not known. So far as limited by temperature, therefore, it is not possible to assign a limit to the descent of water under average conditions of crustal temperature.
But there is reason to think that water does not go down to the depths necessary for the critical temperature. Rock, solid and unyielding as it seems, is yet mobile under sufficiently great pressure. Cracks and cavities are believed to affect it to depths which are slight in comparison with the radius of the earth. If openings were formed at sufficient depths below the surface they could not persist, for the adjacent rock, under the pressure which there exits, would “flow” in and close them. The flow is, in effect (though not in principle) much like the flow of a stiff liquid. The outer zone of the earth where cracks and cavities may exist has been called the zone of fracture,[1] and the thickness of the zone of fracture is not believed to exceed six miles, even for the most resistant rock.[2] This depth is probably much less than that at which the critical temperature of water would be reached, as indicated in the last table.
If water descends through openings in the rock to a depth of six miles, it will, under the assumptions specified in the first and second columns of the following table, have the temperature indicated in the third column:
| Initial Temperature | Rate of Increase of Temperature |
Temperature at Depth of Six Miles |
|---|---|---|
| 50° | 1° for 50 feet | 683° Fahr. |
| 50° | 1° for 75 " | 472° " |
| 50° | 1° for 100 " | 367° " |
In the first of these cases, the temperature of the water at the assumed lower limit of the zone of fracture, is above the critical temperature of water. If the assumptions involved in this case are correct, water might descend to the point where it would be converted into water-gas, and in this condition it might be [p. 200] occluded by the hot rock. In the other cases, which involve the more probable assumptions, the critical temperature is not approached closely at a depth of six miles. If pores and cracks do not extend to greater depths, liquid water could not; and since the water at this depth has probably not reached its critical temperature, it must still be in the form of liquid. It would seem, therefore, that the descent of water under ordinary conditions, is limited by the zone of fracture, rather than by temperature.
Some recent experiments suggest that, at high temperatures and under great pressures, water may enter into combination with rock material, with contraction of volume.[3] If this is correct, water (in some form of combination) may perhaps go down below the zone of fracture.
Movement of ground-water.[4] Ground-water is in more or less continual movement. If all the water is pumped out of a well, it soon fills up again by inflow from the sides. Springs and flowing wells also demonstrate the movement of ground-water. Near the surface the movement is primarily downward if the rock through which it passes is equally permeable in all directions; but so soon as the descending water reaches the water-surface, its downward flow is checked, and its movement is partly lateral.
Ground-water moves chiefly by slow percolation, for most of it is not organized into definite streams. Small streams are seen in some caves, and subterranean streams issue as springs in some places; but most streams which issue as springs probably have definite channels for short distances only, before they appear at the surface. It is probable that ground-water frequently flows in considerable quantity along somewhat definite planes, without having open channels. This is the case especially along the sun . of an impervious layer overlain by a porous one. The “reservoirs” from which artesian wells draw their supply are not usually streams or lakes, but porous beds of rock, containing abundant water. As the supply is drawn off at one point, it is renewed by [p. 201] water entering elsewhere. Since the freedom of movement of ground-water is influenced greatly by the porosity of the rock, and since the rock is, on the average, most porous near the surface, the movement of ground-water is, on the whole, greatest near the surface and least at its limit of descent. It follows that while the upper part of the ground-water, especially that above groundwater level, moves somewhat freely, the lower part moves more slowly. It is probable, indeed, that movement in the lower part of the subterranean hydrosphere is extremely slow.
The amount of ground-water. The porosity of surface rocks varies widely, and the porosity of but few has been determined.[5] From such determinations as have been made, it is estimated that the average porosity of the outer part of the lithosphere is somewhere between five and ten percent. If the porosity diminishes regularly to a depth of six miles, where it becomes zero, the average porosity to this depth would be half the surface porosity.[6] An average porosity of two and one-half per cent would mean that the rock contains enough water to form a layer nearly 800 feet deep, if brought out to the surface.[7] For the reason indicated in the second foot-note below, it may be that this figure is too large, even for the land. The porosity beneath the sea is probably less than that beneath the land, so that for the earth, this figure is perhaps too high, and is not to be regarded as a measurement. It is the water beneath the surface which justifies the term hydrosphere, as applied to the waters of the earth.
Fate of ground-water. Most of the water which sinks into the earth reaches the surface again after a longer or shorter journey. Some of it is evaporated from the surface directly; some of it is [p. 202] taken up by plants and is passed by them into the atmosphere; some of it issues in the form of springs; some of it seeps out; some of it is drawn out through wells; and much of the remainder finds its way underground to the sea or to lakes, issuing as springs beneath them. A small portion of the descending water enters into permanent combination with mineral matter. Many minerals are known to take up water, being changed thereby from an anhydrous to a hydrous condition. It does not necessarily follow, however, that the total supply of water is thereby decreasing. Minerals once hydrated may be dehydrated, the water being set free. Furthermore, considerable quantities of water in the form of vapor issue from volcanoes, and volcanic vents often continue to steam long after volcanic action proper has ceased. It is probable that some, and perhaps much of the water issuing from these vents has never been at the surface before. The amount of water reaching the surface of the earth for the first time from volcanoes, may, so far as now known, equal or even exceed the amount consumed in the hydration of minerals.
¶ The Work of Ground-water
Ground-water is ever active. The results of its work are partly chemical and partly mechanical, the former being the more important.
The Chemical Work
The results of the chemical and chemico-physical action of water may be grouped in several more or less distinct categories.
1. The simplest effect is the solution of mineral matter. Pure water will dissolve certain minerals; but the carbon dioxide extracted from the atmosphere, and the products of organic decay extracted from the soil give ground-water a power to dissolve which is not possessed by pure water. The solvent work of ground-water is shown by the fact that all such water, whether it issues as springs or is drawn out through wells, contains mineral matter, while rain water is essentially free from it. The subtraction of soluble matter from rock tends to make it porous, and helps it to decay.
2. One mineral substance in solution may be exchanged for another extracted from the rock. Thus the lime carbonate of a [p. 203] shell imbedded in rock may be removed, molecule by molecule, and some other substance, such as silica, left in its place. When the process is complete, the substance of the shell has been completely removed, though its form and structure are still preserved in the new material which has taken the place of the old. Buried logs are sometimes converted into stone by the substitution of mineral matter for the vegetable tissue. This is petrifaction (Fig. 167). Solution is a necessary antecedent of substitution.
3. The materials which are subtracted from the rock at one point may be added to other rock elsewhere. Thus a third type of change, addition, is effected. Rock may at one time and place be rendered porous by the subtraction of some of its substance, and the openings thus formed may subsequently become the receptacles of deposits from solution. This is exemplified in the stalactitic deposits of many caves. Not uncommonly cracks and fissures are filled with mineral matter deposited by the waters which pass through them. Thus arise veins (Fig. 20) which, for the most part, are cracks and crevices filled by mineral matter brought to them in solution, and precipitated on their walls. Most veins of metallic ores originated in this way.
4. A further series of changes is effected by ground-water when it, or the mineral matter it contains, enters into combination with the mineral matter through which it passes. One of the commonest processes of this sort, hydration, has already been referred to (p. 102) ; but in the development of many of the commoner hydrous minerals, changes other than hydration are involved. These changes result in new mineral combinations, the new minerals being developed out of the old, usually with some additions or subtractions. In the long course of time, changes of this sort may be so great as to change rock radically, both chemically and physically.
Quantitative importance of solution. In general, solution is probably most effective at a relatively slight distance below the surface. In the mantle rock, the materials are usually less soluble than below, for in many places they represent the residuum after the soluble parts of the formation from which they originated were dissolved out. Below this zone the rock contains more soluble [p. 204] matter, and the water, charged with organic matter in its descent through the soil, is in condition to dissolve it. At greater depths the water has become saturated to some extent, and, so far forth, less active. Here, too, the movement is less free. The increased pressure at considerable depths, on the other hand, facilitates solution, which must be understood to take place under proper circumstances in any zone reached by the water.
Calculations have been made which illustrate in a measure the quantitative importance of the solution effected by ground-water, for the mineral matter dissolved in streams is largely from groundwater. In the case of several streams, among them the Thames and the Elbe, careful estimates of the amount of dissolved mineral matter have been made. The Thames drains an area only about one-tenth as large as the State of New York, but it is estimated to carry about 1,500 tons of mineral matter in solution to the sea daily.[8] From the uppermost 20,000 square miles of its drainage basin, the Elbe is estimated to carry yearly about 1,370,000 tons of mineral matter in solution.
These figures make it clear that ground-water is an effective agent in the lowering of land surfaces. It is estimated that something like one-third as much matter is carried to the sea in solution as in the form of sediment, and that by this process alone land areas would be lowered something like one foot in 13,000 years.[9]
The quantitative importance of the solution effected by groundwater is shown in another way. It is probable that most of the salt now in the sea has been taken to it in solution by waters flowing in from the land. The amount of salt is stupendous (p. 289). Furthermore, most of the limestone of the earth has been extracted from sea-water, whither the larger part of it was carried by streams. The aggregate amount of limestone is far greater than the amount of salt in the sea. Some other sorts of rock, such as gypsum, of less importance quantitatively, have had a similar history. The total amount of rock which must have been decomposed to yield such quantities of calcium carbonate, sodium chloride, etc., is far greater than all the rock now above sea-level.
[p. 205]
Deposition of mineral matter from solution. The deposition of material from solution is effected in several ways. (1) It is sometimes deposited by evaporation. This is well shown where water seeps out on arid lands. (2) Reduction of temperature often occasions deposition. In general, hot water is a better solvent of mineral matter than cold,[10] and if it issues with abundant mineral matter in solution, the precipitation of some of it is likely to take place. (3) Plants sometimes cause the precipitation of mineral matter from solution. About some hot springs, even where the temperature of the water is very high, small plants of low type (algae) grow in profusion. In ways which are not perfectly understood, these algae extract the mineral matter from the hot water. They are now thought to be a chief factor in the deposits about the hot springs of the Yellowstone Park.[11] The influence of organisms on precipitation from solution is not confined to the waters of hot springs. (4) A fourth factor involved in the deposition of mineral matter from solution is relief of pressure. Pressure increases the solvent power of water. It also increases the amount of gas which may be dissolved, and the gas affects the solvent power of the water. As water charged with gas comes to the surface, the pressure is relieved and some of the gas escapes. Such mineral matter as was held in solution by the help of the gas which escapes, is then precipitated. (5) Precipitation is also sometimes effected by the mingling of waters containing different mineral substances in solution. Such mingling of solutions is most common along lines of ready subterranean flow, and while each portion of the water entering a crevice or porous bed might have been able to keep its own mineral matter in solution, their mingling may involve chemical changes, resulting in the formation of insoluble compounds, and therefore in deposition. This principle has probably been involved in the filling of many fissures and crevices, converting them into veins. (6) The disturbance of water, as by waves, some [p. 206] times causes its gases to escape, and so causes the deposition of mineral matter held in solution.
The deposition of material held in solution is most notable at two zones, one below that of most active solution, and the other at the surface, where evaporation is active. Under proper conditions, however, deposition may take place at any level reached by water.
Mechanical Work
The mechanical work of ground-water is relatively unimportant. Where it flows in definite streams, the channels through which it flows are likely to be increased by mechanical erosion as well as by solution. Either beneath the surface, or after the streams issue, the mechanical sediment carried will be deposited.
¶ Results of the Work of Ground-water
Weathering. Where the solvent work of ground-water is slight and equally distributed, the result is to make the rock porous. If, for example, some of the cement of sandstone is dissolved, the texture of the rock becomes more open; but if all the cement is removed, the rock is changed from sandstone to sand. If a complex crystalline rock contains among its many minerals some one which is more soluble than the others, that one may be dissolved. This has the effect of breaking up the rock, since each mineral acts as a binder for the rest. It might happen that no one of the minerals is dissolved completely, but that some one of them is decomposed by water, and certain of its constituents removed. Such change would be likely to cause the mineral so affected to crumble, and with its crumbling, if it is an important constituent of the rock. the integrity of the rock is destroyed. The increase in volume attendant on hydration, etc., sometimes leads to the disruption of rock. These are phases of weathering.
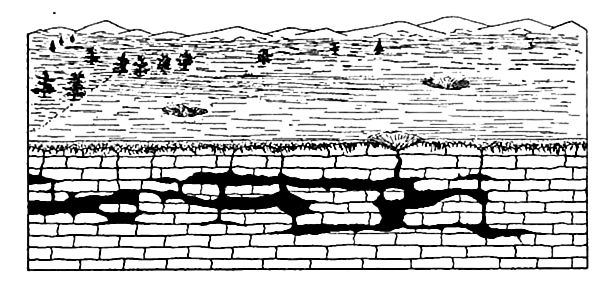
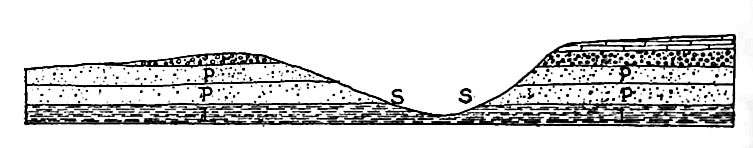
Caverns.[12] Where local solution is very great, results of another sort may be effected. In formations like limestone, which are relatively soluble, considerable quantities of material are frequently [p. 207] dissolved from a given place. Instead of making the rock porous, in the usual sense of the term, large caverns may be developed (Fig. 166). In their production, solution may be abetted by the mechanical action of the water passing through the openings which solution has developed.

[p. 208]
One of the best known regions of caves is in the basin of the Ohio in Kentucky and southern Indiana, where the number of caves is large, and the size of some of them, such as Mammoth and Wyandotte, very great. A ground-plan of Wyandotte (Ind.) Cave is shown in Fig. 168. The aggregate length of its passageways is a number of miles.
Deposition often takes place in caves after they are formed (Fig. 169), or it may even go on at the same time that the cave is being excavated. Stalactites and stalagmites are common forms of cave deposits. A stalactite may start from a drop of water leaking through the roof of the cave. Evaporation, or the escape of some of the carbonic gas in solution, results in the deposition of some of the lime carbonate about the margin of the drop, in the form of a ring. Successive drops make successive deposits on the lower edge of the ring, which grows downward into a hollow tube through which descending water passes, making its chief deposits at the end. Deposition in the tube may ultimately close it, while deposition on the outside, due to the water trickling down in that position, may greatly enlarge it.
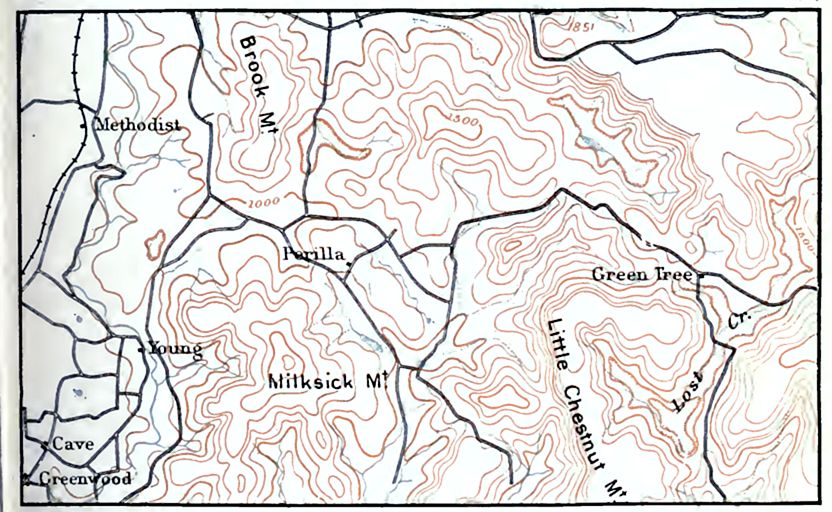
Limestone sinks. Underground caves sometimes give rise to topographic features of local importance. When the roof of a cavern becomes thin and weak, it may collapse, giving rise to a sink or depression in the surface over the site of the original cave. This is so common that regions of limestone caves are often affected by numerous sinks formed in this way. They are conspicuous in the cave region of Kentucky, and are well known in many other limestone districts, and are known as limestone sinks (Fig. 170 and Fig. 166, and PI. XV).
[p. 209]
Creep, slumps, and landslides. When the soil and subsoil on a slope become charged with water, they tend to move downward. When the movement is too slow to be sensible it is called creep; when rapid enough to be sensible, the material is said to slump or slide. This may happen when the slope on which water-charged mantle-rock lies is steep (Fig. 171). Some landslides have done great damage. Where a stream’s banks are high, and of unindurated material, such as clay, considerable masses sometimes slump from the bank or bluff into the river, or settle away slowly from their former positions. This is a common phenomenon along streams which have cut valleys in drift, and along shores of drift on which waves are encroaching. The same phenomenon is common on a larger scale on the slopes of steep mountains.[13]

In creep and in landslides gravity is the force involved, and the ground-water only a condition which makes gravity effective.
[p. 210]
Summary
All in all, ground-water is to be looked upon as a most important geological agent. When it is remembered that a very large part of all the water which falls on the surface of the earth, either in the form of rain or snow, sinks beneath the surface; that much of it sinks to a great depth; that much of it has a long underground course before it reappears at the surface; that it is everywhere and always active, either in subtracting from the rock through which it passes, in adding to it, in effecting the substitution of one mineral substance for another, or in bringing about new chemical combinations; and when it is remembered that these processes have been going on for untold millions of years, it will be seen thai the total result accomplished must be great. The rock formations of the earth to the depths to which ground-water penetrates arc to be looked upon as a sort of chemical laboratory through which waters are circulating in all directions, charged with all sorts of mineral substances. Some of the substances in solution are ticposited beneath the surface, and some are brought to the surface where the waters issue. Much of the material brought to the surface in solution is carried to the sea and utilized by marine organisms in the making of shells. Without the mineral matter brought to the sea by springs and rivers, many shell-bearing animals of great importance, geologically, would perish. Biologically, therefore, as well as geologically, ground-water is of great importance.

[p. 211]
¶ Springs
Springs. The term spring is applied to any water which issues from beneath the surface with sufficient volume to cause a distinct current. If the water issues so slowly as to merely keep the surface moist, it is not called a spring, but seepage. The spring from which water issues with a strong current, especially if it is upward, is comparable to a flowing well, while the spring from which water issues with little force, and without upward movement, is comparable to the flow of water into a common well.

Springs often issue from the sides of valleys (Fig. 172), the bottoms of which are below ground-water level. They are especially likely to issue at the surface of relatively impervious layers, [p. 212] and where the valley slopes cut joints, porous beds, or other structures which allow free flow of ground-water.
Springs are classified in various ways, and the several classifications suggest characteristics worthy of note. They are sometimes classed as deep and shallow, but the idea involved in this grouping would be better expressed by strong and feeble. Springs are also classified as cold and thermal, the latter term meaning simply that the temperature is such as to make the springs seem warm or hot. The temperature of thermal springs ranges up to the boiling-point, of water. Between deep springs and shallow ones, and between cold springs and warm ones, respectively, there is no sharp line of demarkation. Again, some springs are continuous in their flow, while others are intermittent. Most intermittent springs flow after periods of precipitation, but dry up during droughts. Springs are also classified as mineral and common. Mineral springs, in the popular sense of the term, are of two types: (1) Those which contain an unusual amount of mineral matter, and (2) those which contain some unusual mineral. All springs which are not mineral are common. This classification is not very significant, for all springs contain more or less mineral matter, and many springs which are common contain more mineral matter than some springs that are mineral. Mineral springs are themselves classified according to the kind and amount of mineral matter they contain. Thus saline springs contain salt; sulphur springs contain compounds (especially gaseous) of sulphur; chalybeate springs contain iron compounds, especially the sulphate; calcareous springs contain abundant lime carbonate, etc. These various mineral substances have been extracted from the rock. The salt of saline springs is usually extracted from beds of salt beneath the surface. Lime carbonate, one of the commonest substances in solution in ground-water, is dissolved from limestone, or derived by chemical change from rocks containing some calcium compound. Thus lime feldspars, by carbonation, give rise to lime carbonate. Chalybeate waters often arise from the oxidation of iron sulphide (a mineral which is common in many sedimentary rocks) and the solution of the resulting sulphate. Medicinal springs are those which contain some substance or substances which have, or are supposed to have, curative properties.
[p. 213]
Mineral matter in solution. The number and variety of mineral substances in spring water is very great, and the amount of solid matter in solution varies widely. Some of the hot springs of the Yellowstone Park contain nearly three grams (2.8733) of mineral matter per kilogram.[14] The springs of Leuk (Switzerland) bring to the surface annually more than 2,000 tons of calcium sulphate (gypsum) in solution, and in the same time the springs of Bath (England) bring up an amount of mineral matter in solution sufficient to make a column 9 feet in diameter, and 140 feet high.[15]
Geysers. Geysers are intermittently eruptive hot springs. They occur only in volcanic regions (past or present), and in but few of them, being known only in the Yellowstone National Park, Iceland, and New Zealand. There are said to be more than sixty active geysers in the Yellowstone Park.
The cause of the eruption is steam. The surface-water sinks down until, at some unknown depth, it comes into contact with rock sufficiently hot to boil it. The source of the heat is not open to inspection, but it is believed to be the uncooled part of extruded or intruded lava. From what was said earlier in this chapter it is clear that geysers do not have their origin in water which sinks down to the zone of great heat, where the downward increment of heat is normal.
The water of a geyser issues through a tube of unknown length. Whether the tube is open down to the source of the heat is not determinable, but water from such a source finds its way to the tube.
[p. 214]
Water may enter the tube from all sides and at various levels. The heating may precede or follow its entrance into the tube, or both. So far as the water is heated after it enters the tube, the point of most rapid heating may be at the bottom of the tube, or at some point above. If the temperature of the source of heat were high enough to convert the descending water into steam as fast as it enters the tube, the steam would escape continuously, and there would be no geyser; but if the rock is only hot enough to bring the water to the boiling-point after some lapse of time, and after some water has accumulated, an eruption is possible.

The exact sequence of events which leads up to an eruption is not known, but a definite conception of the principles involved may perhaps be secured by a definite case. Suppose a geyser-tube full of water and heated at its lower end. As the water is heated below, convection tends to distribute the heat throughout the column [p. 215] of water above. If convection were free and the tube short, the result would be a boiling spring; but if the tube is long, and especially if convection is impeded, the water at some level below the surface may be brought to the boiling-point earlier than at the top. Under these circumstances if even a little water in the lower part of the tube is converted into steam, the steam will raise the column of water above, and it will overflow. The overflow relieves the pressure on all parts of the column of water below the surface. If before the overflow there was any considerable volume of water essentially ready to boil, the relief of pressure following the overflow might allow it to be converted into steam suddenly, and the sudden conversion of any considerable quantity of water into steam would cause the eruption of all the water above it (Fig. 173). The height to which the water would be thrown depends upon the amount of steam, the size and straightness of the tube, etc.
 .jpg)
It is clear that everything which impedes convection in the geyser tube will hasten the period of eruption, since impeded circulation will have the effect of holding the hot water down, and so of bringing the water at some level below the top more quickly to the boiling-point. It follows that anything which chokes the [p. 216] tube, or which increases the viscosity of the water, hastens an eruption.[16]
Geysers often build up crater-like basins or cones (Figs. 174 to 176) about themselves, the cone being of material deposited from solution. In the Yellowstone Park, the precipitation of the matter in solution (chiefly silica) is partly due to cooling, but largely to the algae which abound even in the boiling water, and the brilliant colors of some of the deposits about the springs are attributable to these plants. When the water from any geyser or hot spring ceases to flow, the plants die and the colors disappear. The heating of geyser water must cool the lava or other source of heat below. As this takes place, the time between eruptions becomes longer and longer. In the course of time, therefore, the geyser must cease to be eruptive, and when this change is brought about the geyser becomes a hot spring. Within historic time several geysers have ceased to erupt and new ones have been developed. In the Yellowstone Park, where there are said to be something like '3,000 vents of all sorts, hot springs which are not eruptive greatly outnumber the geysers. From many of the vents but little steam issues, and from some, little else.

[p. 217]


[p. 218]
A few geysers have somewhat definite periods of eruption. Of such “Old Faithful” is the type; but even this geyser, which formerly erupted at regular intervals of about an hour, is losing the reputation on which its name is based. Not only is its period of eruption lengthening, but it is becoming irregular, and the irregularity appears to be increasing. In the short time during which this geyser has been under observation its period has changed from a regular one of 60 minutes, or a little less, to an irregular one of 60 to 90 minutes. In the case of some geysers, years elapse between eruptions, and in some the date of the last eruption is so remote that it is uncertain whether the vent should be looked upon as a geyser or merely a hot spring.
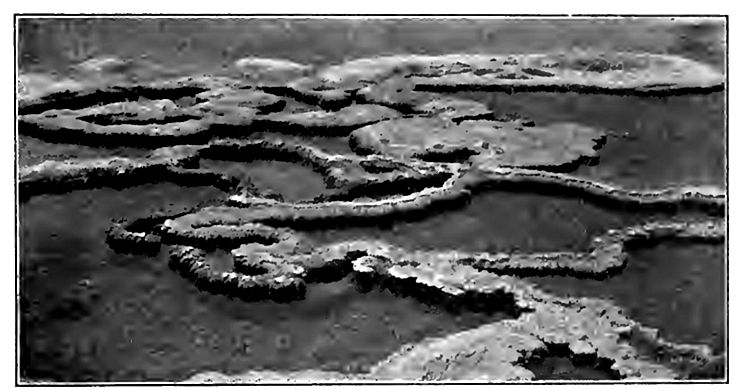
In the Yellowstone Park the geysers are mainly in the bottoms of valleys (Fig. 179), but the deposits characteristic of geysers are found in not a few places well above the present bottoms. These deposits record the fact that in earlier times the geysers were at higher levels than now. It is probable that they have been, at all stages in their history, near the bottoms of the valleys, and thai as the valleys have deepened, the ground-water has found lower [p. 219] and lower points of issue. In this respect the geysers probably have had the same history as other springs.
Unless new intrusions of lava occur, or unless heat is otherwise renewed at the proper points, it is probable that all existing geysers will become extinct within a time which is, geologically, short. New geyser regions may, however, develop as old ones disappear.
Artesian Wells
The terms artesian wells and flowing wells were synonymous originally; but any notably deep well is now called artesian. The artesian well which does not flow does not differ from common wells in principle, while the flowing well is really a gushing spring, the opening of which was made by man.

Flowing wells[17] depend upon certain relations of rock structure, water supply, and elevation. Generally speaking, a flowing well is possible in any place underlain by any considerable bed of porous [p. 220] rock, if this rock outcrops at a sufficiently higher level in a region of adequate rainfall, and is covered by a layer or bed of impervious or relatively impervious rock. This statement involves four conditions, all of which are illustrated by Fig. 180, where a is the bed of porous rock. It is not necessary that the beds of rock form a structural basin, nor is it usually necessary to take account of the character of the rock beneath the porous bed which contains the water.
The bed of porous rock is the “reservoir” of the flowing well. Formations of sand or sandstone, and of gravel or conglomerate, most commonly serve as the reservoirs. In order that they may contain abundant water they must have some thickness, and their outcropping edges must be so situated that the water may enter freely and be replenished, chiefly by rain, as the water flows out at the well.
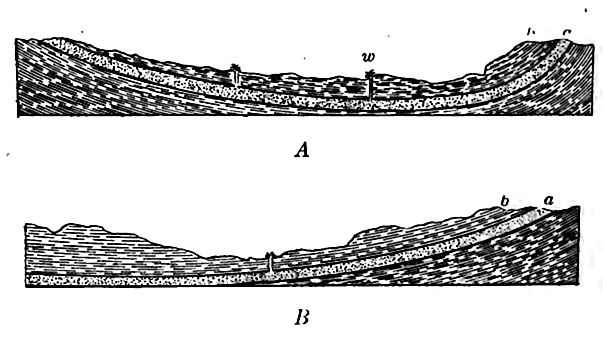
A relatively impervious layer of rock above the reservoir (b.Fig. 180) is most important; otherwise the water in the reservoir will leak out, and there will be little or no “head” at the well site. Thus if the rock overlying stratum a (Fig. 180) were badly broken, the fractures extending up to the surface, the conditions would be unfavorable for flowing wells, for though wells in the positions of those in Fig. 180 might get abundant water, they would not be likely to flow. If the stratum next below the reservoir is not impervious [p. 221], some lower one probably is. No layer of rock is more impervious than one which is full of water, and the substructure of any bod which might serve as a reservoir is usually full of water, even when the rock, if free from water, would be porous.
If the outcrop of the reservoir is notably above the site of the well, and if it is kept full by frequent rains, the “head” will be strong, though the water at the well will not rise to the level of the outcrop of the reservoir. Experience has shown that an allowance of about one foot per mile of subterranean flow should be made. Thus if the site of the well is 100 miles from the outcrop of the waterbearing stratum, and 200 feet below it, the water will rise something like 100 feet above the surface at the well. This rule is, however, not applicable everywhere. The failure of the water to rise to the level of its head is due chiefly to the friction of flow through the rock. The more porous the rock the less the friction. The height of the flow is also influenced by the number of wells drawing on the same reservoir, on the degree of imperviousness of the confining bed above, etc. Flowing wells, often relatively shallow, are frequently obtained from unconsolidated drift.

[p. 222]
Map work. Illustrations of the topographic effects of ground-water may be found on a few topographic maps. Plates XC to XCIV of Professional Paper 60, U. S. Geological Survey, are examples. Various reports on mining regions, showing structure sections, afford illustrations of the economic significance of one phase of work done by ground-water.
¶ Notes
Hoskins. 16th Ann. Rept., U. S. Geol. Surv., p. 853. ↩︎
Van Hise. Principles of North American Pre-Cambrian Geology, 16th Ann. Rept., U. S. Geol. Surv. ↩︎
Barus. Bull. 92, U. S. Geol. Surv. ↩︎
For a full discussion of this subject see King, 19th Ann. Rept., U. S. Geol. Surv., Pt. II, and Slichter, Water Supply and Irrigation Paper No. 67, CJ. S. Geol. Surv. ↩︎
For tables see Buckley, Building and Ornamental Stones, Bull. IV, Wis. Surv.; Merrill, Stones for Building and Decoration; and various Survey Reports. ↩︎
It is probable that the porosity decreases in more than an arithmetic ratio, both because the deeper rocks are not so generally of porous kinds, as at the surface, and because of the pressure which tends to close openings. ↩︎
Slichter (op. cit., p. 15) estimates that the ground-water is sufficient in amount to cover the earth’s surface to a depth of 3,000 to 3,500 feet. Earlier estimates (Delesse, Bull. Soc. Geol., France, Second Series, Vol. XIX, 186162, p. 64) gave still higher figures. Fuller, in a recent estimate, places the amount much lower, about 100 feet. Water Supply and Irrigation Paper 160, U. S. Geol. Survey. ↩︎
Geikie. Text-book of Geology, 3d ed., p. 378. ↩︎
Reade. Liverpool Geol. Soc, 1876 and 1884. ↩︎
This is not true in the case of minerals, such as lime carbonate, dissolved under the influence of gases in solution in the water. ↩︎
Weed. The Formation of Hot Springs Deposits; Excursion to the Rocky Mountains, and Ninth Ann. Rept. U. S. Geol. Surv., pp. 613-76; and B. M. Davis. Science, Vol. VI, pp. 145-57, 1897. ↩︎
For a racy and interesting account of caverns see Shaler’s Aspects of the Earth. ↩︎
Russell has emphasized this point in 20th Ann. U. S. Geol. Surv., Pt. ix, pp. 193-202, and Cross, 21st Ann. U.S. Geol. Surv., Pt. II, pp. 129-150. ↩︎
Gooch and Whitfield. Bull. 47, U. S. Geol. Surv. ↩︎
Geikie. Text-book of Geology, 3d ed., p. 367. ↩︎
Weed. Ninth Ann. Rept. U. S. Geol. Surv., pp. 613-70, and Am. .lour. Sci., Vol. XXXVII, 1889, pp. 351-59. ↩︎
Chamberlin. Geol. of Wis., Vol. I, pp. 689-97, and Fifth Ann. Rept., U. S. Geol. Surv., pp. 131-73. The former a brief, and the latter an elaborate, exposition of the principles involved. The same principles in various forms of statement have appeared in later publications of the U. S. Geol. Surv. ↩︎